��Ŀ����
����Ŀ������˵������ȷ����
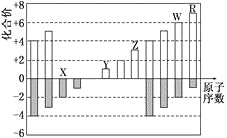
A. ��֪t1��ʱ����ӦC+CO2![]() 2CO ��H >0������Ϊ�����������¶ȣ��淴Ӧ���ʼ�С
2CO ��H >0������Ϊ�����������¶ȣ��淴Ӧ���ʼ�С
B. ��ѹ�����з�����ӦN2+O2![]() 2NO�����������г���He�����淴Ӧ���ʾ�����
2NO�����������г���He�����淴Ӧ���ʾ�����
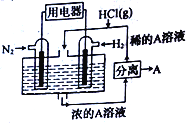
C. ��һ������п�ۺ�����6molL-1���ᷴӦʱ��Ϊ�˼�����Ӧ���ʣ��ֲ�Ӱ�����H2������������Ӧ���м���������CuSO4��Һ
D. ���ڹ�ҵ�ϳɰ���ӦN2+3H2![]() 2NH3��ѡ��450����ҪΪ����ߴ���������ý���Ĵ�Ч��
2NH3��ѡ��450����ҪΪ����ߴ���������ý���Ĵ�Ч��
���𰸡�D
��������A������¶ȣ���ѧ��Ӧ���ʼӿ죬�����淴Ӧ���ʾ��ӿ죬��A����B��ں�ѹ�����з�����ӦN2+O2![]() 2NO�����������г���He������������Ӧ���������Ũ�ȼ�С�����淴Ӧ�����ʶ���С����B����C���һ������п�ۺ�����6molL-1���ᷴӦʱ����Ӧ���м���������CuSO4��Һ��п������ͭ��Ӧ����ͭ������ԭ���ԭ�����ӿ��˻�ѧ��Ӧ���ʣ������������������٣���C����D���ҵ�ϳɰ�Ϊ���ȷ�Ӧ��ѡ��450����¶ȿ϶�����Ϊ�����ת���ʣ�����Ϊ����߷�Ӧ���ʣ�û�в��ø��ߵ��¶���Ҫ����450��ʱ����������ý���Ĵ�Ч����ã���D��ȷ��
2NO�����������г���He������������Ӧ���������Ũ�ȼ�С�����淴Ӧ�����ʶ���С����B����C���һ������п�ۺ�����6molL-1���ᷴӦʱ����Ӧ���м���������CuSO4��Һ��п������ͭ��Ӧ����ͭ������ԭ���ԭ�����ӿ��˻�ѧ��Ӧ���ʣ������������������٣���C����D���ҵ�ϳɰ�Ϊ���ȷ�Ӧ��ѡ��450����¶ȿ϶�����Ϊ�����ת���ʣ�����Ϊ����߷�Ӧ���ʣ�û�в��ø��ߵ��¶���Ҫ����450��ʱ����������ý���Ĵ�Ч����ã���D��ȷ��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�����Ŀ����������һ����Ҫ�����ȼ�ϣ�Ҳ�������������������ȣ��Գ��������ƻ����á���ҵ�Ͽ�����ú���������ˮú�����ϳɶ����ѡ�
��ش��������⣺
��1��ú����������Ҫ��ѧ��Ӧ����ʽΪ��___________________________��
��2��ú�����������в������к�����H2S��Na2CO3��Һ���գ�����������ʽ�Σ��÷�Ӧ�Ļ�ѧ����ʽΪ��________________________________________��
��3������ˮú���ϳɶ����ѵ�������Ӧ���£�
�� 2H2(g) + CO(g) ![]() CH3OH(g)����H �� ��90.8 kJ��mol��1
CH3OH(g)����H �� ��90.8 kJ��mol��1
�� 2CH3OH(g) ![]() CH3OCH3(g) + H2O(g)����H�� ��23.5 kJ��mol��1
CH3OCH3(g) + H2O(g)����H�� ��23.5 kJ��mol��1
�� CO(g) + H2O(g) ![]() CO2(g) + H2(g)����H�� ��41.3 kJ��mol��1
CO2(g) + H2(g)����H�� ��41.3 kJ��mol��1
�ܷ�Ӧ��3H2(g) + 3CO(g) ![]() CH3OCH3(g) + CO2 (g)����H�� ___________��
CH3OCH3(g) + CO2 (g)����H�� ___________��
һ�������µ��ܱ������У����ܷ�Ӧ�ﵽƽ�⣬Ҫ���CO��ת���ʣ����Բ�ȡ�Ĵ�ʩ��__________������ĸ���ţ�
a�����¸�ѹ b��������� c������CO2��Ũ��
d������CO��Ũ�� e�������������
��4����֪��Ӧ��2CH3OH(g) ![]() CH3OCH3(g) + H2O(g)ij�¶��µ�ƽ�ⳣ��Ϊ400 �����¶��£����ܱ������м���CH3OH ����Ӧ��ijʱ�̲�ø���ֵ�Ũ�����£�
CH3OCH3(g) + H2O(g)ij�¶��µ�ƽ�ⳣ��Ϊ400 �����¶��£����ܱ������м���CH3OH ����Ӧ��ijʱ�̲�ø���ֵ�Ũ�����£�
���� | CH3OH | CH3OCH3 | H2O |
Ũ��/��mol��L��1�� | 0.44 | 0.6 | 0.6 |
�� �Ƚϴ�ʱ�����淴Ӧ���ʵĴ�С��v�� ______ v�� ������>������<����������)��
�� ������CH3OH��10 min��Ӧ�ﵽƽ�⣬��ʱc(CH3OH) �� _________����ʱ���ڷ�Ӧ����v(CH3OH) �� __________���ﵽƽ��ʱ�״���ת��������CH3OH��= _________