ЬтФПФкШн
ЁОЬтФПЁПЕЊдЊЫиЪЧвЛжжживЊЕФЗЧН№ЪєдЊЫиЃЌПЩаЮГЩЖржжЛЏКЯЮяЁЃЪдЛиД№ЯТСагаЙиЮЪЬтЃК
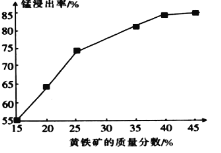
ЃЈ1ЃЉЂйвбжЊ4CO(g)ЃЋ2NO2(g) ![]() 4CO2(g)ЃЋN2(g) ІЄHЃНЃ1 200 kJЁЄmolЃ1ЖдгкИУЗДгІЃЌИФБфФГвЛЗДгІЬѕМўЃЌЃЈвбжЊЮТЖШT2>T1ЃЉЯТСаЭМЯёе§ШЗЕФЪЧ_______(ЬюДњКХ)
4CO2(g)ЃЋN2(g) ІЄHЃНЃ1 200 kJЁЄmolЃ1ЖдгкИУЗДгІЃЌИФБфФГвЛЗДгІЬѕМўЃЌЃЈвбжЊЮТЖШT2>T1ЃЉЯТСаЭМЯёе§ШЗЕФЪЧ_______(ЬюДњКХ)

ЂквбжЊCOгыH2OдквЛЖЈЬѕМўЯТПЩвдЗЂЩњЗДгІЃКCO(g)ЃЋH2O(g)![]() CO2(g)ЃЋH2(g)ЁЁІЄH= Ѓ Q kJЁЄmolЃ1 ЃЌ820 ЁцЪБдкМзЁЂввСНИіКуШнУмБеШнЦїжаЃЌЦ№ЪМЪБАДееЯТБэНјааЭЖСЯЃЌОЙ§вЛЖЮЪБМфКѓДяЕНЦНКтзДЬЌЃЌШєМзжаCOЕФзЊЛЏТЪЮЊ40%ЃЌдђИУЗДгІЕФЦНКтГЃЪ§ЮЊ____________ЃЛввШнЦїЮќЪеЕФШШСПЮЊ________________ЁЃ
CO2(g)ЃЋH2(g)ЁЁІЄH= Ѓ Q kJЁЄmolЃ1 ЃЌ820 ЁцЪБдкМзЁЂввСНИіКуШнУмБеШнЦїжаЃЌЦ№ЪМЪБАДееЯТБэНјааЭЖСЯЃЌОЙ§вЛЖЮЪБМфКѓДяЕНЦНКтзДЬЌЃЌШєМзжаCOЕФзЊЛЏТЪЮЊ40%ЃЌдђИУЗДгІЕФЦНКтГЃЪ§ЮЊ____________ЃЛввШнЦїЮќЪеЕФШШСПЮЊ________________ЁЃ
![]()
ЃЈ2ЃЉыТПЩзїЮЊЛ№М§ЗЂЖЏЛњЕФШМСЯЃЌгыбѕЛЏМСN2O4ЗДгІЩњГЩN2КЭЫЎеєЦјЃЌвбжЊЃК
Ђй N2 (g) +2O2 (g) = N2O4 (l) ІЄH =Ѓ19.5 kJЁЄmolЃ1
Ђк N2H4(l)+O2(g) = N2(g) +2H2O(g) ІЄH =Ѓ534.2 kJЁЄmolЃ1
аДГіыТгыЫФбѕЛЏЖўЕЊЗДгІЕФШШЛЏбЇЗНЪН______________ЁЃ
ЃЈ3ЃЉДПЕФЕўЕЊЫс(HN3)ЪЧЮоЩЋвКЬхЃЌГЃгУзів§БЌМСЃЌГЃЮТЯТЯђ25 mL 0.1molЁЄLЃ1 NaOHШмвКжаМгШы0. 2molЁЄLЃ1 HN3ЕФШмвКЃЌЕЮМгЙ§ГЬжаЕФpHжЕЕФБфЛЏЧњЯпЃЈШмвКЛьКЯЪБЕФЬхЛ§БфЛЏКіТдВЛМЦЃЉШчЭМЁЃ

ЂйИљОнЭМЯёаДГіHN3ЕФЕчРыЗНГЬЪН:_________ЁЃ
ЂкЯТСаЫЕЗЈе§ШЗЕФЪЧ_________________ЃЈЬюађКХЃЉ
AЃЎШєгУвбжЊХЈЖШЕФNaOHШмвКЕЮЖЈHN3ШмвКРДВтЖЈHN3ЕФХЈЖШЪБгІгУМзЛљГШзїжИЪОМС
BЃЎГЃЮТЯТЃЌЯђ0.2 molЁЄLЃ1 HN3ЕФШмвКжаМгЫЎЯЁЪЭЃЌдђ![]() ВЛБф
ВЛБф
CЃЎЗжБ№жаКЭpHОљЮЊ4ЕФHN3ШмвККЭHClШмвКЃЌЯћКФ0.1 molЁЄLЃ1 NaOHШмвКЕФЬхЛ§ЯрЭЌ
DЃЎDЕуЪБШмвКжаРызгХЈЖШДцдкШчЯТЙиЯЕЃК2c(H+) + c(HN3) =c(N3Ѓ) + 2c(OHЃ)
ЁОД№АИЁПвв ![]() 0.12Q kJ 2N2H4(l) + N2O4 (l) = 3N2 (g) + 4H2O (g) ІЄІЇ=-1048.9 kJЁЄmol-1 HN3
0.12Q kJ 2N2H4(l) + N2O4 (l) = 3N2 (g) + 4H2O (g) ІЄІЇ=-1048.9 kJЁЄmol-1 HN3![]() H++N3ЈD BD
H++N3ЈD BD
ЁОНтЮіЁП
ЃЈ1ЃЉЂйИљОнгАЯьЛЏбЇЗДгІЫйТЪЕФвђЫиЃКЮТЖШЁЂХЈЖШЁЂбЙЧПЁЂДпЛЏМСРДШЗЖЈЛЏбЇЗДгІДяЦНКтгУЕНЕФЪБМфЃЌИљОнгАЯьЛЏбЇЦНКтвЦЖЏЕФвђЫиЃКЮТЖШЁЂХЈЖШЁЂбЙЧПРДШЗЖЈЛЏбЇЦНКтжаИїИіСПЕФБфЛЏЧщПіЃЛ
ЂкИљОнЛЏбЇЦНКтШ§ЖЮЪНСаЪНвдМАЛЏбЇЗДгІжаCOЕФзЊЛЏТЪЮЊ40%НјааМЦЫуМДПЩЃЛ
ЃЈ2ЃЉвРОнИЧЫЙЖЈТЩЃЌНсКЯЬтИЩШШЛЏбЇЗНГЬЪНМЦЫуаДГіЁЃ
ЃЈ3ЃЉЂйвРОнЭМЯѓПЩжЊЕўЕЊЫсЪєгкШѕЫсЃЌОнДЫЪщаДЕчРыЗНГЬЪНМДПЩЁЃ
ЂкAЃЎЕЮЖЈжеЕуЪБЃЌШмвКгЩЫсадБфЮЊМюадЃЌОнДЫбЁдёжИЪОМСЃЛ
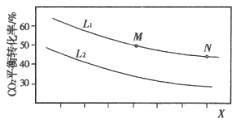
BЃЎвРОнKжЛгыЮТЖШгаЙиНјааЗжЮіЃЛ
CЃЎвРОнШѕЫсДцдкЕчРыНјааЗжЮіЃЛ
DЃЎвРОнЮяСЯЪиКуКЭЕчКЩЪиКуХаЖЯЁЃ
ЃЈ1ЃЉЂйМзЃКЩ§ИпЮТЖШЃЌЛЏбЇЦНКтФцЯђвЦЖЏЃЌе§ЁЂФцЗДгІЫйТЪЛсбИЫйдіДѓЃЌЛсРыПЊдРДЕФЫйТЪЕуЃЌЙЪМзДэЮѓЃЛввЃКЩ§ИпЮТЖШЃЌЛЏбЇЗДгІЫйТЪдіДѓЃЌЫљвдT2ЪБЯШДяЕНЛЏбЇЦНКтзДЬЌЃЌВЂЧвЛЏбЇЦНКтФцЯђвЦЖЏЃЌЖўбѕЛЏЕЊЕФзЊЛЏТЪМѕаЁЃЌЙЪвве§ШЗЃЛБћЃКЖдгкЗДгІЃК4CO(g)ЃЋ2NO2(g) 4CO2(g)ЃЋN2(g)ЃЌTВЛБфЃЌдіДѓбЙЧПЃЌЦНКте§ЯђвЦЖЏЃЌвЛбѕЛЏЬМЕФЬхЛ§ЗжЪ§ЛсМѕаЁЃЌЙЪБћДэЮѓЃЌЙЪбЁввЁЃ
ЂквбжЊМзжаCOЕФзЊЛЏТЪЮЊ40%ЃЌдђгаЃК
CO(g)ЃЋH2O(g)![]() CO2(g)ЃЋH2(g)
CO2(g)ЃЋH2(g)
ГѕЪМСПЃК0.1 0.1 0 0
БфЛЏСПЃК0.04 0.04 0.04 0.04
ЦНКтСПЃК0.1-0.04 0.1-0.04 0.04 0.04
ЩшШнЦїЕФШнЛ§ЮЊVLЃЌдђK=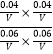 =
=![]() ЁЃ
ЁЃ
дкКуЮТКуШнЬѕМўЯТЃЌввКЭМзЛЅЮЊЕШаЇЦНКтЃЌЦНКтГЃЪ§ЯрЭЌЃЌМД
CO(g)ЃЋH2O(g)![]() CO2(g)ЃЋH2(g)
CO2(g)ЃЋH2(g)
ГѕЪМСПЃК0 0 0.2 0.2
БфЛЏСПЃКx x x x
ЦНКтСПЃКx x 0.2- x 0.2- x
дђK=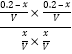 =
=![]() ЃЌНтЕУx=0.12ЃЌЙЪввШнЦїЮќЪеЕФШШСПЮЊ0.12Q kJЁЃ
ЃЌНтЕУx=0.12ЃЌЙЪввШнЦїЮќЪеЕФШШСПЮЊ0.12Q kJЁЃ
ЃЈ2ЃЉЂй N2 (g) +2O2 (g) = N2O4 (l) ІЄH1=Ѓ19.5 kJЁЄmolЃ1
Ђк N2H4(l)+O2(g) = N2(g)+2H2O(g) ІЄH2 =Ѓ534.2 kJЁЄmolЃ1
ИљОнИЧЫЙЖЈТЩЂкЁС2-ЂйЕУЕНыТКЭЫФбѕЛЏЖўЕЊЗДгІЕФШШЛЏбЇЗНЪНЮЊ2N2H4(l) + N2O4 (l) = 3N2 (g) + 4H2O (g) ІЄІЇ=-1048.9 kJЁЄmol-1ЁЃ
ЃЈ3ЃЉЂйвРОнЭМЯѓПЩжЊЕўЕЊЫсЪєгкШѕЫсЃЌВЛФмЭъШЋЕчРыЃЌгУПЩФцКХЃЌЙЪЕчРыЗНГЬЪНЮЊЃКHN3![]() H++N3-ЃЌЙЪД№АИЮЊЃКHN3
H++N3-ЃЌЙЪД№АИЮЊЃКHN3![]() H++N3-ЁЃ
H++N3-ЁЃ
ЂкAЃЎЕЮЖЈжеЕуЪБЃЌШмжЪЮЊЕўЕЊЫсФЦШмвКЃЌЕўЕЊЫсИљРызгЫЎНтЯдМюадЃЌШмвКгЩЫсадБфЮЊМюадЃЌгІбЁгУЗгЬЊЪдвКзїжИЪОМСЃЌЙЪAДэЮѓЁЃ
BЃЎГЃЮТЯТЃЌЯђ0.2 molL-1HN3ЕФШмвКжаМгЫЎЯЁЪЭЃЌЫЎЕФРызгЛ§ГЃЪ§ВЛБфЃЌЕчРыЦНКтГЃЪ§ВЛБфЃЌМДгаЃКKw=c(H+)ЁСc(OH-)ЃЌKa=![]() ЃЌKa=
ЃЌKa= = KwЁСimg src="http://thumb.1010pic.com/questionBank/Upload/2019/05/03/08/785509a5/SYS201905030836124054790767_DA/SYS201905030836124054790767_DA.007.png" width="85" height="45" style="-aw-left-pos:0pt; -aw-rel-hpos:column; -aw-rel-vpos:paragraph; -aw-top-pos:0pt; -aw-wrap-type:inline" />ЃЌKaВЛБфЃЌKwВЛБфЃЌЙЪ
= KwЁСimg src="http://thumb.1010pic.com/questionBank/Upload/2019/05/03/08/785509a5/SYS201905030836124054790767_DA/SYS201905030836124054790767_DA.007.png" width="85" height="45" style="-aw-left-pos:0pt; -aw-rel-hpos:column; -aw-rel-vpos:paragraph; -aw-top-pos:0pt; -aw-wrap-type:inline" />ЃЌKaВЛБфЃЌKwВЛБфЃЌЙЪ![]() ВЛБфЃЌЙЪBе§ШЗЁЃ
ВЛБфЃЌЙЪBе§ШЗЁЃ
CЃЎpHжЕЯрЕШЕФбЮЫсгыЕўЕЊЫсжавбОЕчРыГіЕФЧтРызгХЈЖШЯрЭЌЃЌДЫЪБЯћКФЧтбѕЛЏФЦШмвКЯрЭЌЃЌЕЋЪЧгЩгкЕўЕЊЫсЪЧШѕЫсЃЌМЬајЕчРыГіЕФЧтРызгЛЙашвЊЧтбѕЛЏФЦНјаажаКЭЃЌЙЪЕўЕЊЫсЯћКФЕФЧтбѕЛЏФЦЕФЬхЛ§ДѓЃЌЙЪCДэЮѓЁЃ
DЃЎDЕугЩЕШЮяжЪЕФСПХЈЖШЕФNaN3КЭHN3зщГЩЕФЛьКЯвКЃЌвРОнЮяСЯЪиКуЃК2cЃЈNa+ЃЉ=cЃЈN3-ЃЉ+cЃЈHN3ЃЉЃЌКЭЕчКЩЪиКуcЃЈNa+ЃЉ+cЃЈH+ЃЉ=cЃЈN3-ЃЉ+cЃЈOH-ЃЉЃЌПЩЕУ2c(H+) + c(HN3) =c(N3Ѓ) + 2c(OHЃ)ЃЌЙЪDе§ШЗЁЃ

 дФЖСПьГЕЯЕСаД№АИ
дФЖСПьГЕЯЕСаД№АИЁОЬтФПЁПДХадВФСЯВњвЕЪЧ21ЪРМЭИїЙњОКЯрЗЂеЙЕФИпПЦММжЇжљВњвЕжЎвЛЃЌзїЮЊаХЯЂВњвЕКЭЛњЕчЙЄвЕЕФживЊЛљДЁЙІФмВФСЯЃЌДХадВФСЯЙуЗКгІгУгкЕчзгаХЯЂЁЂОќЪТММЪѕЕШСьгђЁЃЬМЫсУЬжївЊгУгкжЦБИШэДХЬњбѕЬхЃЌЙЄвЕЩЯвдШэУЬПѓ(жївЊГЩЗжЮЊMnO2)КЭЛЦЬњПѓ(жївЊГЩЗжЮЊFeS2)ЮЊжївЊдСЯжЦБИЬМЫсУЬЕФжївЊЙЄвеСїГЬШчЯТЃК

вбжЊЃКМИжжН№ЪєРызгГСЕэЕФpHШчЯТБэЁЃ
Fe2ЃЋ | Fe3ЃЋ | Cu2ЃЋ | Mn2ЃЋ | |
ПЊЪМГСЕэЕФpH | 7.5 | 3.2 | 5.2 | 8.8 |
ЭъШЋГСЕэЕФpH | 9.2 | 3.7 | 7.8 | 10.4 |
ЛиД№ЯТСаЮЪЬтЃК
(1)ЮЊСЫЬсИпШмНўЙЄађжадСЯЕФНўГіаЇТЪЃЌВЩШЁЕФДыЪЉВЛКЯРэЕФга________ЁЃ
AЃЎНСАш BЃЎЪЪЕБЩ§ИпЮТЖШ
CЃЎбаФЅПѓЪЏ DЃЎМгШызуСПЕФеєСѓЫЎ
(2)ШмНўЙ§ГЬжажївЊВњЩњЕФРызгЮЊFe3ЃЋЁЂMn2ЃЋЁЂSO42-ЃЌЧыаДГіжївЊЗДгІЕФРызгЗНГЬЪНЃК___________ЃЛНўШЁКѓЕФШмвКжаКЌгаЩйСПFe2ЃЋЁЂCu2ЃЋЁЂCa2ЃЋЃЌдђдкМгШыЪЏЛвЕїНкШмвКЕФpHДгЖјЪЙЬњдЊЫиБЛЭъШЋГСЕэЧАЃЌМгШыЪЪСПЕФШэУЬПѓФПЕФЪЧ______________________ЃЌМгШыЪЏЛвЕїНкШмвКpHЕФЗЖЮЇЮЊ____________________ЁЃ
(3)ОЛЛЏЙЄађЕФФПЕФЪЧГ§ШЅШмвКжаЕФCu2ЃЋЁЂCa2ЃЋЕШдгжЪЃЌЙЪТЫдќжївЊЪЧ________(ЬюЛЏбЇЪН)КЭCaF2ЁЃШєВтЕУТЫвКжаc(FЃ)ЮЊ0.01molЁЄLЃ1ЃЌдђТЫвКжаВаСєc(Ca2ЃЋ)ЮЊ________molЁЄLЃ1[вбжЊЃКKsp(CaF2)ЃН1.46ЁС10Ѓ10]ЁЃ
(4)гаШЫШЯЮЊОЛЛЏЙЄађжаЪЙгУNaFЛсв§Ц№ЮлШОЃЌНЈвщгУ(NH4)2CO3ДњЬцNaFЃЌЕЋЪЧгУ(NH4)2CO3ДњЬцNaFЕФШБЕуЪЧ______________________________________ЁЃ
(5)ШчЭМЮЊЛЦЬњПѓЕФжЪСПЗжЪ§ЖдУЬНўГіТЪЕФгАЯьЃЌНіОнЭМжааХЯЂЃЌЛЦЬњПѓЕФжЪСПЗжЪ§гІБЃГждк________%зѓгвЁЃ