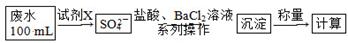��Ŀ����
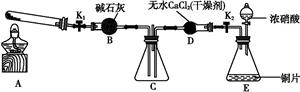
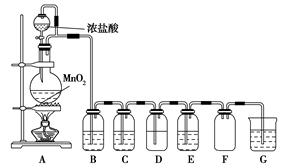
ij��ѧ��ȤС����ľ̿��Ũ����Ϊ��ʼԭ�ϣ�̽��һ��������������Ʒ�Ӧ�Ʊ��������ơ����װ������(����װ���п�����Ӱ��)����ش��������⣺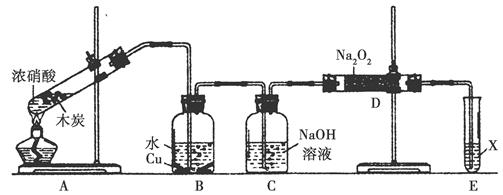
��1����װ������������е�һ�������_________________��
��2��װ��A���Թ��з�����Ӧ�Ļ�ѧ����ʽ��_______________��
��3���Ʋ�B�п��Թ۲쵽����Ҫ������________��Cװ�õ�������________��
��4��װ��D�г�����NaNO2�⣬������һ�ֹ�̬����Y��Y�Ļ�ѧʽ��________������ͨ���ʵ��Ľ���������Y���ʣ���������Ľ�������______________________��
��5����֪�������������ᣬ���ȶ��������´��ڷ�Ӧ3HNO2=HNO3��2NO����H2O����������Һ�У�NO2-�ɽ�MnO4-��ԭΪMn2�������������ɡ�
��д������D�в������������Ƶķ�����_________________��
��Eװ�����Լ�X������________��
| A��ϡ���� | B�����Ը��������Һ |
| C��ϡ���� | D��ˮ |
��1�����װ�õ�������
��2��C��4HNO3(Ũ)  CO2��4NO2����2H2O
CO2��4NO2����2H2O
��3��ͭƬ���ܽ⣬��Һ������������ɫ���ݡ���ȥNO�л��е�CO2
��4��NaOH����װ�м�ʯ�ҵĸ���ܴ���Cװ��
��5���ٽ������������Թ��У�����ϡ���ᣬ��������ɫ���岢��Һ���Ϸ���Ϊ����ɫ����D�в�������������(�������������Թ��У���������KMnO4��Һ������Һ��ɫ��ȥ����D�в�������������)����B
����

 ���ƽ̸�������ѡ����ĩ���100��ϵ�д�
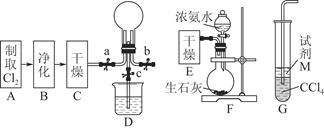
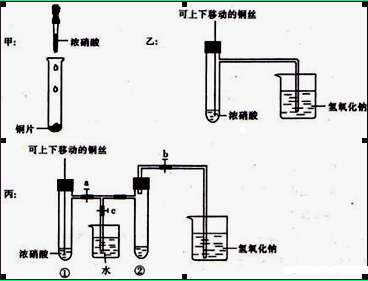
���ƽ̸�������ѡ����ĩ���100��ϵ�д�ij��ȤС����Ƴ���ͼ��ʾװ�����Ľ��̲��С�ͭ�����ᷴӦ��ʵ�飬��̽����ѧʵ�����ɫ����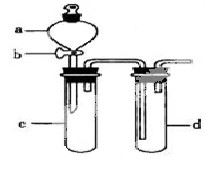
��1��ʵ��ǰ���رջ���b���Թ�d�м�ˮ����û�����ܿڣ������Թ�c��d�Ľ���������c����Ŀ����__________��
��2����d�м�����NaOH��Һ��c�з�һС��ͭƬ���ɷ�Һ©��a��c�м���2mLŨ���ᣬc�з�Ӧ�Ļ�ѧ����ʽ��______________________��
����a��c�м�2mL����ˮ��c�е�ʵ��������_____________��
��3���±�����ȡ����ͭ�����ַ�������������ɫ��ѧ�������ѷ�����_____��������_______��
| ���� | ��Ӧ�� |
| �� | Cu��Ũ���� |
| �� | Cu��ϡ���� |
| �� | Cu��O2��ϡ���� |
���ü�������������ȡ����Ӧ��ȡ����Ʒ����������ڹ�ҵ���ѳ�Ϊ��ʵ��ij��ѧ��ȤС����ʵ������ģ���������̣�����Ƶ�ģ��װ�����£�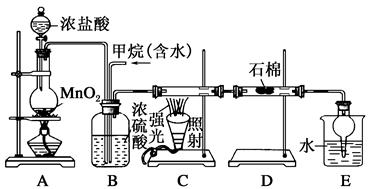
�������Ҫ��ش�
��1��Bװ�������ֹ��ܣ��ٿ��������ٶȣ��ھ��Ȼ�����壻��______________��
��2����V��Cl2��/V��CH4����x��������������������Ȼ��⣬��xֵӦ________��
��3��Dװ�õ�ʯ���о��Ȼ���KI��ĩ����������____________��
��4��Eװ�õ�������________������ţ���
| A���ռ����� | B���������� |
| C����ֹ���� | D�������Ȼ��� |
��6��Eװ�ó����������⣬�������л����E�з�����������ѷ���Ϊ________����װ�û���ȱ�ݣ�ԭ����û�н���β����������β����Ҫ�ɷ�Ϊ________������ţ���
a��CH4��b��CH3Cl��c��CH2Cl2��d��CHCl3��e��CCl4
��ҵβ���е�������ͨ�����ð������շ�����ԭ����NH3��NOx�ڴ��������·�Ӧ�����������ʡ�ijУ�С��ͬѧ��������װ�úͲ���ģ�ҵ�ϵ�������Ĵ������̡�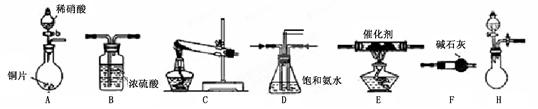
I��̽����ȡNH3�ķ���
��1��������װ���У�H�ܿ��١������ȡNH3��װ������Ҫ���ӵķ�Ӧ�Լ�Ϊ ��
��2��Ϊ̽�����õ�ʵ��Ч�����С��ͬѧ��������Cװ������ȡ�������ڿ���ʵ��������ͬ������£�����±���ʵ�����ݡ�
| �Լ������� | �����Լ� | NH3�����mL�� | |
| a | 6.0 g Ca(OH)2�������� | 5.4 g NH4Cl | 1344 |
| b | 5.4g (NH4)2SO4 | 1364 | |
| c | 6.0 g NaOH�������� | 5.4 g NH4Cl | 1568 |
| d | 5.4g (NH4)2SO4 | 1559 | |
| e | 6.0 g CaO�������� | 5.4 g NH4Cl | 1753 |
| f | 5.4 g (NH4)2SO4 | 1792 | |
�����������ݣ�����Ϊ���ַ�����ȡ������Ч����� ������ţ����Ӹ÷���ѡ���ԭ�Ϸ�������Ч���õĿ���ԭ���� ��
II��ģ��β������
�С��ͬѧѡ����������װ�ã�������˳�����ӳ�ģ��β������װ�ý���ʵ�顣
��1���������װ����ѡ������Ϊ�����Ľ��в��䣨��ѡװ�ò����ظ�����
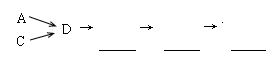
��2��A�з�Ӧ�����ӷ���ʽΪ ��
��3��Dװ�õ������У�ʹ�����Ͼ��ȡ����������ٶȡ� ��
��4��Dװ���е�Һ�廹�ɻ��� ������ţ���
a��H2O b��CCl4 c��ŨH2SO4 d��CuSO4��Һ
��5����С��ͬѧ����Ƶ�ģ��β������װ���л�����һ�����Ե�ȱ���� ��
 SiHCl3��H2
SiHCl3��H2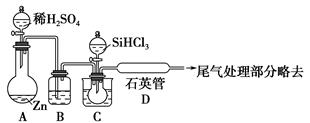
 NaNO3+NaNO2+H2O
NaNO3+NaNO2+H2O