��Ŀ����
10�� ���ú˴Ź������ⶨ�л�����ӵ���ά�ṹ���о������2002��ŵ������ѧ�������л�������У���ͬλ�õ���ԭ�ӵĺ˴Ź������и����ķ�ֵ���źţ�Ҳ��ͬ�����ݷ�ֵ���źţ�����ȷ���л����������ԭ�ӵ��������Ŀ��
���ú˴Ź������ⶨ�л�����ӵ���ά�ṹ���о������2002��ŵ������ѧ�������л�������У���ͬλ�õ���ԭ�ӵĺ˴Ź������и����ķ�ֵ���źţ�Ҳ��ͬ�����ݷ�ֵ���źţ�����ȷ���л����������ԭ�ӵ��������Ŀ����1�����з����У���˴Ź���������ֻ��һ�ַ壨�źţ���������AD��
A��CH3CH3 B��CH3COOH C��CH3COOCH3 D��CH3COCH3
��2��������A��B�ķ���ʽ����C2H4Br2��A�ĺ˴Ź���������ͼ��ʾ����A�Ľṹ��ʽΪCH2BrCH2Br����Ԥ��B�ĺ˴Ź�����������2���壨�źţ�
��3��ij�л����������̼���⡢������Ԫ����ɣ����ⶨ����Է�������Ϊ90����ȡ���л�����Ʒ1.8g����������������ȫȼ�գ��������Ⱥ�ͨ��Ũ����ͼ�ʯ�ң����߷ֱ�����1.08g��2.64g��
��������л���ķ���ʽC3H6O3��Ҫ�м�����̣���
�ڸ��л���ͨ��������ײ�ú����Ȼ����ǻ������ţ���˴Ź�����������4�ַ壬�����֮��Ϊ3��1��1��1����д�����л���Ľṹ��ʽCH3CH��OH��COOH��
���� ��1���˴Ź��������з�ֵ�������л�������ԭ�ӵ����������˴Ź���������ֻ����һ�ַ壬˵���÷����е�Hԭ�Ӷ��ǵ�Ч�ģ�ֻ��1��Hԭ�ӣ�
��2����A�ĺ˴Ź�������֪��������ֻ��һ��Hԭ�ӣ�A������2��Brԭ�����Ӳ�ͬ��̼ԭ�ӣ�B��A��Ϊͬ���칹�壬B������2��Brԭ��������ͬһ̼ԭ���ϣ���Ϸ�����Hԭ�������жϺ˴Ź�������ͼ��������ֵ��
��3��Ũ��������Ϊȼ������ˮ����������ʯ������Ϊ���ɶ�����̼������������n=$\frac{m}{M}$�����л��ˮ��������̼�����ʵ������ٸ���ԭ���غ���������C��Hԭ����Ŀ�������Է��������ɼ���Oԭ�Ӹ���������ȷ������ʽ����Ϻ��еĹ�������˴Ź�������ȷ���ṹ��ʽ��
��� �⣺��1���˴Ź���������ֻ����һ�ַ壬˵���÷����е�Hԭ�Ӷ��ǵ�Ч�ģ�ֻ��1��Hԭ�ӣ�
A��CH3CH3��6��Hԭ�Ӷ��ǵ�Ч�ģ��˴Ź���������ֻ����һ�ַ壬��A��ȷ��
B��CH3COOH�м��е�Hԭ�����Ȼ��е�Hԭ��������ѧ������ͬ��CH3COOH��2��Hԭ�ӣ��˴Ź�����������2���壬��B����
C��CH3COOCH3�м�������ѧ������ͬ����Hԭ�Ӳ�ͬ����2��Hԭ�ӣ��˴Ź�����������2���壬��C����
D��CH3COCH3��2��������ͬһ���ʻ��ϣ�6��Hԭ�Ӷ��ǵ�Ч�ģ��˴Ź���������ֻ����һ�ַ壬��D��ȷ��
��ѡ��AD��
��2����A�ĺ˴Ź�������֪��������ֻ��һ��Hԭ�ӣ�A������2��Brԭ�����Ӳ�ͬ��̼ԭ�ӣ���A�Ľṹ��ʽΪCH2BrCH2Br��B��A��Ϊͬ���칹�壬B������2��Brԭ��������ͬһ̼ԭ���ϣ�BΪCH3CHBr2����������2��Hԭ�ӣ��ʺ˴Ź�������ͼ��2���壻
�ʴ�Ϊ��CH2BrCH2Br��2��
��3�����л�������ʵ���Ϊ$\frac{1.8g}{90g/mol}$=0.02mol��n��H2O��=$\frac{1.08g}{18g/mol}$=0.06mol��n��CO2��=$\frac{2.64g}{44g/mol}$=0.06mol�������л��������N��C��=$\frac{0.06mol}{0.02mol}$=3��N��H��=$\frac{0.06mol��2}{0.02mol}$=6������Ϊ�л��������Ϊ90�����л�����N��O��=$\frac{90-12��3-6}{16}$=3�����л������ʽΪC3H6O3��
�ʴ�Ϊ��C3H6O3��
�ڸ��л���ͨ��������ײ�ú����Ȼ����ǻ������ţ���˴Ź�����������4�ַ壬�����֮��Ϊ3��1��1��1���л���Ľṹ��ʽΪCH3CH��OH��COOH���ʴ�Ϊ��CH3CH��OH��COOH��
���� ���⿼��˴Ź������ס�ͬ���칹�����д���л������ʽ��ṹʽ��ȷ�����ѶȲ�������Ź����������л�������ԭ�ӵ������ϵ��������ѧ�����������ͼ��������Ŀ��飬��Ŀ�ѶȲ���

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�| A�� | NH3�Ǽ��Է��ӣ���ռ乹��Ϊƽ���������� | |
| B�� | CCl4�ǷǼ��Է��ӣ����е�C-Cl��Ϊ���Թ��ۼ� | |
| C�� | CO2 �ǷǼ��Է��ӣ�����������ԭ������������ˮ�� | |
| D�� | BF3�Ǽ��Է��ӣ�������ԭ�ӵ��ӻ��������Ϊsp2�ӻ� |
| A�� | ���ڱ��д������£�����Ԫ���γɵĵ����۷е���� | |
| B�� | ͬ����Ԫ��ԭ�Ӱ뾶�����Ӱ뾶�ݱ������ͬ | |
| C�� | HF��HCl��HBr��HI���۷е������� | |
| D�� | �������Ӽ��Ļ�����һ�������ӻ����� |
| A�� | ��ͭ | B�� | ��ͭ | C�� | Ӳ�� | D�� | �ƽ� |
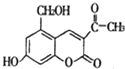 ij�������⾷ҩ����Ч�ɷ��ǡ��������ء�����ṹ��ʽ��ͼ��ʾ�����ڡ��������ء���˵���У���ȷ���ǣ�������
ij�������⾷ҩ����Ч�ɷ��ǡ��������ء�����ṹ��ʽ��ͼ��ʾ�����ڡ��������ء���˵���У���ȷ���ǣ�������| A�� | ���������ء��ķ���ʽΪC12H12O5 | |
| B�� | ���������ء����������ᷢ��������Ӧ | |
| C�� | 1 mol���������ء�����ܺ�2 mol NaOH��Ӧ | |
| D�� | 1 mol���������ء�����ܺ�4 mol H2�����ӳɷ�Ӧ |
| A�� | c��Na+��=2c��CO32-�� | B�� | c��HCO3-����c��H2CO3�� | ||
| C�� | c��Na+����[c��CO32-��+c��HCO3-��] | D�� | c��OH-��=c��H+��+c��HCO3-��+2c��H2CO3�� |
| A�� | ��������ԭ�Ӳ�ȡsp3�ӻ�����ɼ��ķ��������幹�Ͷ������������� | |
| B�� | sp3�ӻ��������ͬһ��ԭ�������������s�����p�����������γɵ�һ������������¹�� | |
| C�� | CH4�����е�sp3�ӻ��������4��Hԭ�ӵ�1s�����Cԭ�ӵ�2p�������������γɵ� | |
| D�� | ��AB3�͵Ĺ��ۻ����������ԭ��A������sp3�ӻ�����ɼ� |
 B��
B��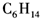 C��
C��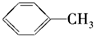 D��
D��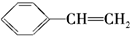
 ���÷���Ҳ������ij���ϵĵ��壬д���ϳɸ����ϵĻ�ѧ����ʽ��
���÷���Ҳ������ij���ϵĵ��壬д���ϳɸ����ϵĻ�ѧ����ʽ�� ��
��