��Ŀ����
�ڸ���ʱ��������Ҫ�ɷ�FeS2����������Ӧ���������������Ͷ����������������е����ʲ����뻯ѧ��Ӧ����ij��ѧ�о�ѧϰС�����������Ʒ��������ʵ��̽����
��ʵ��һ���ⶨ��Ԫ�صĺ���
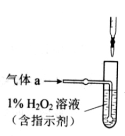
I��ʵ��װ������ͼ��ʾ���гֺͼ���װ����ʡ�ԣ���A�е��Լ���˫��ˮ����m��g����������Ʒ����Ӳ�ʲ�����D�С���A��B����εμ�Һ�壬ʹ���巢��װ�ò��ϵػ����������壬��������Ӳ�ʲ�����D�е���������Ʒ����Ӧ��ȫ��
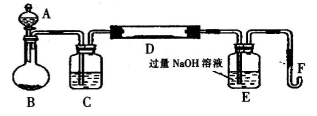
II����Ӧ������Eƿ�е���Һ�������´�����

��ʵ������ⶨ��Ԫ�صĺ���
III���ⶨ��Ԫ�غ�����ʵ�鲽�����£�
��������ϡ�����ܽ�����ȴ��Ӳ�ʲ�����D�еĹ��壬���ˣ��õ���ҺA;
������ҺA�м�������Ļ�ԭ��ʹ��Һ�е�Fe3+��ȫת��ΪFe2+�����ˣ��õ���ҺB��
�۽���ҺBϡ��Ϊ250 mL;
��ȡϡ��Һ25��00 mL,��Ũ��Ϊc mol/L������KMnO4��Һ�ζ������εζ�ʵ������KMnO4��Һ��
����ƽ��ֵΪV mL��
���������ʵ�飬�ش��������⣺
��1��I��װ��C��ʢװ���Լ���_ ;Eƿ��������_ ��
��2��11�м������H2O2��Һ������Ӧ�����ӷ���ʽΪ____��
��3��111�IJ�����У�����ҺBϡ��Ϊ250 mL��Ҫ�õ��IJ����������ձ�������������ͷ�ι��⣬
������Ҫ�õ�����_ ������ʢװKMnO3��Һ��������_ ��
��4������������ʵ���������Ԫ�غ���Ԫ�ض�û����ʧ�������������Ʒ����Ԫ�ص���������Ϊ_ �����ú�m1��m2�Ĵ���ʽ��ʾ������Ԫ�ص���������Ϊ____�����ú�m1��c��v�Ĵ���ʽ��ʾ��
��ʵ��һ���ⶨ��Ԫ�صĺ���
I��ʵ��װ������ͼ��ʾ���гֺͼ���װ����ʡ�ԣ���A�е��Լ���˫��ˮ����m��g����������Ʒ����Ӳ�ʲ�����D�С���A��B����εμ�Һ�壬ʹ���巢��װ�ò��ϵػ����������壬��������Ӳ�ʲ�����D�е���������Ʒ����Ӧ��ȫ��

II����Ӧ������Eƿ�е���Һ�������´�����

��ʵ������ⶨ��Ԫ�صĺ���
III���ⶨ��Ԫ�غ�����ʵ�鲽�����£�
��������ϡ�����ܽ�����ȴ��Ӳ�ʲ�����D�еĹ��壬���ˣ��õ���ҺA;
������ҺA�м�������Ļ�ԭ��ʹ��Һ�е�Fe3+��ȫת��ΪFe2+�����ˣ��õ���ҺB��
�۽���ҺBϡ��Ϊ250 mL;
��ȡϡ��Һ25��00 mL,��Ũ��Ϊc mol/L������KMnO4��Һ�ζ������εζ�ʵ������KMnO4��Һ��
����ƽ��ֵΪV mL��
���������ʵ�飬�ش��������⣺
��1��I��װ��C��ʢװ���Լ���_ ;Eƿ��������_ ��
��2��11�м������H2O2��Һ������Ӧ�����ӷ���ʽΪ____��
��3��111�IJ�����У�����ҺBϡ��Ϊ250 mL��Ҫ�õ��IJ����������ձ�������������ͷ�ι��⣬
������Ҫ�õ�����_ ������ʢװKMnO3��Һ��������_ ��
��4������������ʵ���������Ԫ�غ���Ԫ�ض�û����ʧ�������������Ʒ����Ԫ�ص���������Ϊ_ �����ú�m1��m2�Ĵ���ʽ��ʾ������Ԫ�ص���������Ϊ____�����ú�m1��c��v�Ĵ���ʽ��ʾ��
��14�֣���1��Ũ���ᣨ1�֣������ն�������2�֣���2��H2O2��SO32����SO42����H2O��3�֣�
��3��250ml����ƿ��2�֣�����ʽ�ζ��ܣ�2�֣�
��4��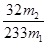 ��100%��2�֣���
��100%��2�֣���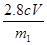 ��100%��2�֣�
��100%��2�֣�
��3��250ml����ƿ��2�֣�����ʽ�ζ��ܣ�2�֣�
��4��
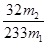 ��100%��2�֣���
��100%��2�֣���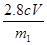 ��100%��2�֣�
��100%��2�֣������������1��A��ʢ��˫��ˮ��˵����װ�����Ʊ������ġ����������к���ˮ����������C��ʢװ���Լ���Ũ���ᣬ����������������������Ʒ�������ڸ����·�Ӧ����SO2������E������������Һ�����������ն�������
��2������������Һ���ն������������������ƣ�����11�м������H2O2��Ŀ���������������ƣ����������ơ������Һ�з�����Ӧ�����ӷ���ʽΪH2O2��SO32����SO42����H2O.
��3��111�IJ�����У�����ҺBϡ��Ϊ250 mL��Ҫ�õ��IJ����������ձ�������������ͷ�ι��⣬
������Ҫ�õ�����250mL����ƿ������ȡϡ��Һ25.00ml����Һ��������Һ���������Ҫ��С�������λ�����õ���������ʽ�ζ��ܣ����Ԣ���ʢװKMnO3��Һ����������ʽ�ζ��ܡ�
��4���������ó��������ᱵ��������ԭ���غ��������������������Ϊx����
S����BaSO4
32g 233g
x m2g
���x��
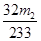 g
g��������������
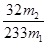 ��100%
��100%������صĻ�ԭ������Mn2�����ڷ�Ӧ�еõ�7��2��5�����ӡ���Fe2���ڷ�Ӧ��ʧȥ1�����ӣ����Ը��ݵ��ӵĵ�ʧ�غ��֪����25.00ml��Һ���������ӵ����ʵ�����cmol/L��0.001VL��5��0.005cVmol��������Ԫ�ص�����������
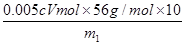 ��100%��
��100%��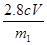 ��100%
��100%
��ϰ��ϵ�д�
 ��ʱѵ���������������ϵ�д�
��ʱѵ���������������ϵ�д�
�����Ŀ