��Ŀ����
����Ŀ����ѧʵ���Ҳ����ķ�Һ�к��д�������Ⱦ���������ʣ�Ϊ�˱�����������ѧ�����Ҳ����ķ�Һ���뾭����������ŷš�ij��ѧʵ���Ҳ��������Է�Һ�к���Fe3+��Cu2+��Ba2+���ֽ��������Ӻ�Cl-�������ӣ�ʵ������������������Է�Һ���д������Ի��ս������ⶨ����������������
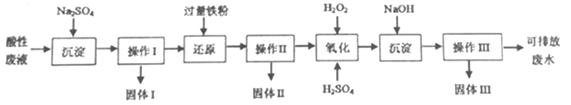
��֪ʵ���д��������Է�Һ�����ΪIL����pH�Ʋⶨ������H+���ʵ���Ũ��Ϊ0.10mol��L-1.
��ش��������⣺.
(1)ʵ���в���I��II��III�ķ�����ͬ��������������Ϊ���ˡ�_________�����
(2)������������������Һ��ɫΪ�ػ�ɫ���÷�Ӧ�����ӷ���ʽΪ��_________
(3)������ŷŵķ�ˮ�в���Fe3+�ķ����ǣ�_________
(4)ʵ���г�������I������Ϊ4.66g������II������Ϊ15.2g����������ϡ�����ܽ����II������˱�״���µ���ɫ����4.48L�������II�н���ͭ������Ϊ_________
(5)ʵ���н�����II���г�����գ�ʹ���������˾ƾ��ơ����żܡ������ǡ��������⣬����_________(������)
���𰸡�ϴ�� 2Fe2++H2O2+2H+=2Fe3++2H2O ȡ���ŷŵķ�ˮ�������μ�KSCN��Һ�������Һ����죬˵�����ŷŵķ�ˮ�в���Fe3+ 9.6 ����
��������
�ں���Fe3+��Cu2+��Ba2+��Cl-�����Է�Һ�еμӹ���Na2SO4��Һ��ͨ������I�Ĺ��ˡ�ϴ�Ӳ�����ɵ�BaSO4����(����I)����Һ�м������Fe�ۿɻ�ԭ��Һ�е�Cu2+��ͨ���������Ĺ��ˡ�ϴ�Ӳ�����ɵ�Fe��Cu�Ļ����(����II)����������Һ�еμӹ�����H2O2��Һ���ɽ���Һ�е�Fe2+����ΪFe3+���μӹ���NaOH��Һ�ɵ�Fe(OH)3����(����III)�����������Ĺ��ˡ�ϴ�Ӳ�����ɵ�Fe(OH)3��������Һ���ŷţ�
(1)��ʵ������Ҫͨ�����ɳ������������ж���Һ�и������ĺ��������Ի�ȡ�����辭���ˡ�ϴ�ӡ����
(2)��������Ϊ˫��ˮ��Fe2+������Fe3+�Ĺ��̣����ӷ���ʽΪ2Fe2++H2O2+2H+=2Fe3++2H2O��
(3)Fe3+���Ժ�KSCN��Һ��Ӧʹ��Һ�Ժ�ɫ�����Կ�������KSCN��Һ��Fe3+���м��飬���巽��Ϊ��ȡ���ŷŵķ�ˮ�������μ�KSCN��Һ�������Һ����죬˵�����ŷŵķ�ˮ�в���Fe3+��
(4)����IIΪFe��Cu�Ļ�������Ϊ15.2g����������ϡ�����ܽ����II�������״���µ���ɫ����NOΪ4.48L�������ʵ���Ϊ![]() =0.2mol����Cu�����ʵ���Ϊx mol��Fe�����ʵ���Ϊy mol����64x+56y=15.2��2x+3y=0.2��3(�����غ�ʽ)����ã�x=0.15��y=0.1����Cu�����ʵ���Ϊ0.15mol����Cu������Ϊ64g/mol��0.15mol=9.6g��
=0.2mol����Cu�����ʵ���Ϊx mol��Fe�����ʵ���Ϊy mol����64x+56y=15.2��2x+3y=0.2��3(�����غ�ʽ)����ã�x=0.15��y=0.1����Cu�����ʵ���Ϊ0.15mol����Cu������Ϊ64g/mol��0.15mol=9.6g��
(5)����������Ҫ�������н��У���ʵ���н�����II���г�����գ�ʹ���������˾ƾ��ơ����żܡ������ǡ��������⣬����������

 ��У����ϵ�д�
��У����ϵ�д�