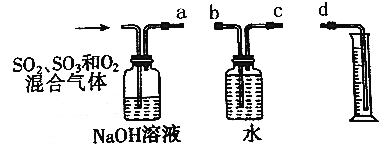
��Ŀ����
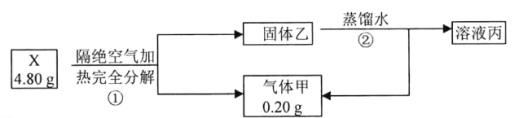
����Ŀ����̼���(��Ҫ�ɷ�ΪCeFCO3)����ȡϡ�������ұ�������Ҫ����ԭ�ϣ��Է�̼���Ϊԭ����ȡ��Ĺ���������ͼ��ʾ���ش��������⣺

(1)CeFCO3��Ce�Ļ��ϼ�Ϊ___________��
(2)��������ʱ����ʹ���մ�������ԭ����_____________________________________��
(3)�������պ�IJ���֮һΪCeO2�������ʱ������Ӧ�����ӷ���ʽΪ____________��
(4)HT��һ��������ˮ���л��ܼ���������������Ϊ_________��
(5)Ϊ���������ʣ������ʵ���߷�Ӧ�¶ȣ����¶�ƫ�߽����ʷ������С����ԭ����_______________________________________��
(6)�л���HT�ܽ�Ce3+��ˮ��Һ����ȡ�������ù��̿ɱ�ʾΪCe3+ (ˮ��)+3HT(�л���) ![]() CeT3 (�л���)+3H+ (ˮ��)����CeT3(�л���)�м���ϡ�����ܻ�ýϴ��ĺ�Ce3+ˮ��Һ����ƽ��ǶȽ�����ԭ��___________________________________��
CeT3 (�л���)+3H+ (ˮ��)����CeT3(�л���)�м���ϡ�����ܻ�ýϴ��ĺ�Ce3+ˮ��Һ����ƽ��ǶȽ�����ԭ��___________________________________��
(7)��֪��298Kʱ��Ksp[Ce(OH)3]=5��l0-20������Һ��c(Ce3+)=0.05mol��L-1���Ӽ����pH��_________ʱCe3+��ʼ����(���ԼӼ��������Һ����仯)��
(8) д����Ce(OH)3����Һ��ͨ�������õ���ƷCe(OH)4�Ļ�ѧ����ʽ��___________��
���𰸡�+3 �մɻ������ɵ�HF��Ӧ 2CeO2+H2O2+6H+ = 2Ce3++O2��+4H2O ��Һ �¶����ߣ�˫��ˮ�����ֽ⣬��ɽ�����ƫС ����Һ�м���ϡ���ᣬʹc(H+)����ƽ�����γ�Ce3+ˮ��Һ�����ƶ� 8 4Ce(OH)3+O2+2H2O = 4Ce(OH)4
��������
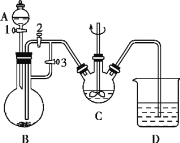
������Ҫ���칤ҵ���̡�����֮ǰ��Ҫ�ȹ۲칤������ͼ����ȥ������е�ÿһ��ȥ����������Ŀ�ļ�ע������پݴ˴��⡣
��1��CeFCO3��CO32�������ԣ�2�ۣ�F�ԣ�1�ۣ��������ϼ۴�����Ϊ0���Ƴ�Ce�ļ�̬Ϊ��3�ۣ�
��2���մ������к���SiO2��SiO2��HF������Ӧ��SiO2��4HF=SiF4����2H2O����˸�������������ʱ����ʹ���մ�������
��3���л�����CeF3��Ce�ԣ�3�ۣ�CeO2��Ce�ԣ�4�ۣ��������ʱ��Ce�ļ�̬���ͣ�CeO2Ϊ��������H2O2Ϊ��ԭ�������ӷ���ʽΪ2CeO2��H2O2��6H��=2Ce3����O2����4H2O��
��4������I�õ���Һ���л��࣬��˲���I������Ϊ��Һ��
��5���������̣����ʱ����H2O2��H2O2�ڸ����·����ֽ⣬H2O2��CeO2������ӦʱH2O2�������٣���ɽ����ʽ��ͣ�
��6���ù����д���Ce3+ (ˮ��)+3HT(�л���) ![]() CeT3 (�л���)+3H+ (ˮ��)������ϡ���ᣬc(H��)����ƽ�����淴Ӧ������У�����Ce3����ˮ��Һ���ܻ�ýϴ���Ce3��ˮ��Һ��
CeT3 (�л���)+3H+ (ˮ��)������ϡ���ᣬc(H��)����ƽ�����淴Ӧ������У�����Ce3����ˮ��Һ���ܻ�ýϴ���Ce3��ˮ��Һ��
��7��Ce3����ʼ���ֳ�����c(Ce3��)��c3(OH��)=Ksp[Ce(OH)3]������c(OH��)=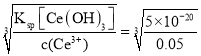 mol��L��1=1��10��6mol��L��1������pH=8ʱCe3����ʼ���ֳ�����
mol��L��1=1��10��6mol��L��1������pH=8ʱCe3����ʼ���ֳ�����
��8������������Ce(OH)3������Ce(OH)4���䷴Ӧ�ķ���ʽΪ4Ce(OH)3��O2��2H2O=4Ce(OH)4��

����Ŀ��ij������Һ�к����±������е�һ�ֻ���֡��ֱ�ȡ����ˮ��Һ���ݽ���ʵ�飬������£�
������ | Fe2+��Fe3+��Al3+ |
������ | SO32-��CO32-��SiO32-��I����NO3- |
����һ����Һ�м���������ᣬ�������ݣ���Һ��ɫ������Գ��壻
�ڼ��������Һ�м����������Ȼ�̼�������ã��²�����Ϻ�ɫ���ϲ���Һ���ֻ�ɫ��
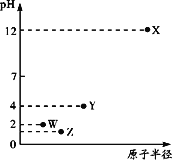
������һ����Һ�м����������������Һ����������������������Һ�������ϵ��ͼ��ʾ��
����������Ϣ���ش��������⣺
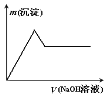
��1��ԭ��Һ�п϶����е�������______________________________________________��
��2���������ɵ�������______________(�ѧ����)�������е�������_____________������ĸ��ţ���
A����ɫ��ζ B���ܱ�NaOH��Һ���� C�����ڴ�����Ⱦ�� D��������ˮ
��3�����з�����Ӧ�����ӷ���ʽ��_______________________________________________��
��4�����г����ܽ�Ļ�ѧ��Ӧ����ʽ��________________________________________��