��Ŀ����
����Ŀ��Ԫ�����ڱ���ѧϰ��ѧ����Ҫ����,������������Ϣ���ɡ��±����������ֶ�����Ԫ�ص�ԭ�Ӱ뾶����Ҫ���ϼ�(��֪��Ԫ�ص�ԭ�Ӱ뾶Ϊ0.089 nm)��
Ԫ�ش��� | A | B | C | D | X | Y |
ԭ�Ӱ뾶/nm | 0.037 | 0.143 | 0.102 | 0.099 | 0.074 | 0.075 |
��Ҫ���ϼ� | +1 | +3 | +6,-2 | -1 | -2 | +5,-3 |
��1��CԪ�������ڱ��е�λ��Ϊ________�������ӽṹʾ��ͼΪ��_______��
��2��B������������Ӧ��ˮ������Y������������Ӧ��ˮ���ﷴӦ�����ӷ���ʽΪ___��
��3������C��D ����Ԫ��˵����ȷ����__________������ţ���
a.�����ӵİ뾶D>C b.��̬�⻯����ȶ���D��Cǿ c.����������Ӧ��ˮ���������C��Dǿ
��4����100mL18mol/L��C������������Ӧ��ˮ�����Ũ��Һ�м��������ͭƬ������ʹ���ַ�Ӧ��������������Ϊ6.72L(����£�����÷�Ӧ������ת�Ƶĵ�����Ϊ______��
��5��д����A��D��X����Ԫ����ɵ�ij�ֿ�������ɱ�����ʵĵ���ʽ_____________��
��6���Ƚ�YԪ������ͬ�������Ԫ�ص��⻯����۷е�ߵ�__ ��___ (���⻯�ﻯѧʽ��������___________��
���𰸡���������VIA��  Al(OH)3+3H+==Al3++3H2O b 0.6NA
Al(OH)3+3H+==Al3++3H2O b 0.6NA  NH3 PH3 ���߶�Ϊ���Ӿ��壬NH3���ڷ��Ӽ��������ǿ���Ӽ��������������۷е��PH3��
NH3 PH3 ���߶�Ϊ���Ӿ��壬NH3���ڷ��Ӽ��������ǿ���Ӽ��������������۷е��PH3��
��������
����Ԫ�ص�ԭ�Ӱ뾶��Դ�С����Ҫ���ϼ������жϳ�Ԫ�����࣬Ȼ����Ԫ�������ɺ�������ʵ����ʷ������
A����Ҫ���ϼ���+1�ۣ�ԭ�Ӱ뾶��С��A��H��C����Ҫ���ϼ���+6�ͣ�2�ۣ����C��S��B����Ҫ���ϼ���+3�ۣ�ԭ�Ӱ뾶�������B��Al��D��X����Ҫ���ϼ۷ֱ��ǣ�1�ͣ�2�ۣ������ԭ�Ӱ뾶���ж�D��Cl��X��O��Y����Ҫ���ϼ���+5�ͣ�3�ۣ�ԭ�Ӱ뾶������ԭ�Ӷ�С����ԭ�ӣ���Y��N��
��1��CԪ����S�������ڱ��е�λ��Ϊ��������VIA�壬�����ӽṹʾ��ͼΪ ��
��
��2��B������������Ӧ��ˮ������������������Y������������Ӧ��ˮ�������ᷴӦ�����ӷ���ʽΪAl(OH)3+3H+��Al3++3H2O��
��3��a.�����Ӻ������Ӿ���18���ӣ��˵����Խ�����Ӱ뾶ԽС��������ӵİ뾶D��C��a����
b.�ǽ�����Cl��S���ǽ�����Խǿ���⻯��Խ�ȶ�������̬�⻯����ȶ���D��Cǿ��b��ȷ��
c.�ǽ�����Խǿ����ۺ����������Խǿ��������������Ӧ��ˮ���������D��Cǿ��c����
��ѡb��
��4��Ũ�����ͭ��Ӧ��������ͭ�����������ˮ����Ԫ�ػ��ϼ۴�+6�۽��͵�+4�ۣ��õ�2�����ӡ���Ӧ�����ɶ�����������ʵ�����6.72L��22.4L/mol��0.3mol����ת�Ƶ��ӵ����ʵ�����0.6mol��������0.6NA��
��5����H��O��Cl����Ԫ����ɵ�ij�ֿ�������ɱ�������Ǵ����ᣬ�����ʽΪ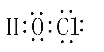 ��
��
��6�����ڰ��������ⶼΪ���Ӿ��壬NH3���ڷ��Ӽ��������ǿ���Ӽ����������Ӷ������۷е��PH3��

����Ŀ���������̺��ŷḻ����Դ�����ú�ˮ��ȡ��ˮ�����þ�IJ��ֹ�����ͼ��ʾ��

��1����ˮ�����ķ�����_________________�����ӽ������͵��������ȡ�
��2���ô�������Ũ����ˮ������Br2:
3Br2+ 6Na2CO3+3H2O�� 5NaBr+ NaBrO3+6NaHCO3��������3 mol Br2ʱ����Ӧ��ת�Ƶĵ�������_____________��
��3����ˮ�в������ӵĺ������£�
�ɷ� | ������ mg/L�� | �ɷ� | ������ mg/L�� |
Na+ | 10560 | Cl- | 18980 |
Mg2+ | 1272 | Br- | 64 |
Ca2+ | 400 | SO42- | 2560 |
���У�Mg2+�����ʵ���Ũ����____________����Ҫ��100 L�ĺ�ˮ����ȡþ����������Ҫ���������Ca��OH��2��������_______________��
��4����Mg��OH��2�Ƶ���Mg�����·�������ʵ���________________������ţ���
A��Mg��OH��2 ![]() MgO
MgO![]() Mg
Mg
B��Mg��OH��2![]() MgO
MgO![]() Mg
Mg
C��Mg��OH��2![]() ��ˮMgCl2
��ˮMgCl2![]() Mg
Mg
D��Mg��OH��2 ![]() MgCl2��Һ
MgCl2��Һ![]() Mg
Mg
����Ŀ��һ���¶��£����ݻ�Ϊ2L���ܱ������з�����ӦCO(g)+H2O(g) ![]() CO2(g)+H2(g)���������ݼ��±�������t2>t1��
CO2(g)+H2(g)���������ݼ��±�������t2>t1��
��Ӧʱ�䣯min | n(CO)��mol | n(H2O)��mol | n(CO2)��mol | n(H2)��mol |
0 | 1.20 | 0.60 | 0 | 0 |
t1 | 0.80 | |||
t2 | 0.20 |
����˵����ȷ����
A. ��Ӧ��t1minĩ������Ϊv(H2)=0.40��t1 mol��L-1��min-1
B. ƽ��ʱCO��ת����Ϊ66��67��
C. ���¶��·�Ӧ��ƽ�ⳣ��Ϊ1
D. �����������䣬����ʼʱ��n(CO)=0.60 mol��n(H2O)=1.20 mol����ƽ��ʱn(CO2)=0.20mol
