��Ŀ����
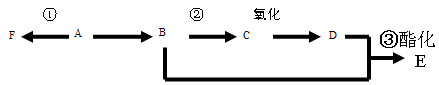
��18�֣�����2.8g�л���A����ȫȼ������0.15molCO2��1.8gH2O��A������ͼ����ͼ��ʾ����֪��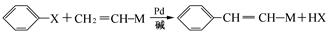 ��XΪ±ԭ�ӣ�MΪ������������ȡ������)�����л���A�ϳ�G���㶹�أ��IJ������£�
��XΪ±ԭ�ӣ�MΪ������������ȡ������)�����л���A�ϳ�G���㶹�أ��IJ������£�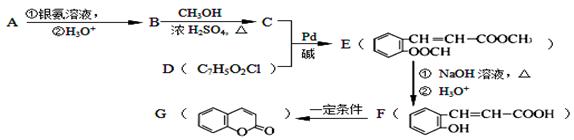
�ش��������⣺
��1��A�ķ���ʽΪ ��
��2��д��C�к�������������:�� ����F��G �ķ�Ӧ������ ��
��3��д��A��������Һ��Ӧ�Ļ�ѧ����ʽ�� ����
��4��D�Ľṹ��ʽΪ�� ����
��5�������㶹�أ�  ���������㶹�ص����Ʒ����������㶹�غ�����һ��ͬ���칹�壨
���������㶹�ص����Ʒ����������㶹�غ�����һ��ͬ���칹�壨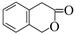 ����Ҫ�õ����Լ��У�NaOH��Һ���� ����
����Ҫ�õ����Լ��У�NaOH��Һ���� ����
��6��F�ж���ͬ���칹�壬д��ͬʱ������������������ͬ���칹��Ľṹ��ʽ�� ����
��. �����г������⣬��������״�ṹ�� ��.���������������ڶ�λ��ȡ������
��. �ܷ���ˮ�ⷴӦ��������Na��Ӧ�� ��.��������Cu(OH)2�����ʵ�����1:2����������Ӧ
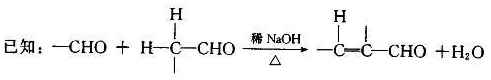
��7����֪��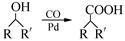 (R��R��Ϊ����)����д���Ա��ͱ�ϩ��
(R��R��Ϊ����)����д���Ա��ͱ�ϩ��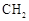 ��CH��CH3��Ϊԭ�ϣ��ϳ�
��CH��CH3��Ϊԭ�ϣ��ϳ�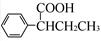 ��·������ͼ���£�
��·������ͼ���£�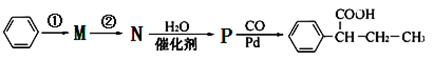
����ٵķ�Ӧ�������Լ�____________������ڵķ�Ӧ����____________��P�Ľṹ��ʽ ��
(1) C3H4O��2�֣� (2)������1�֣� ȡ����Ӧ/������Ӧ��1�֣�
(3) CH2��CHCHO +2Ag(NH3)2OH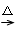 CH2��CHCOONH4 +2Ag��+ 3NH3 +H2O��2�֣�
CH2��CHCOONH4 +2Ag��+ 3NH3 +H2O��2�֣�
(4)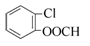 ��2�֣�(5)ϡ���ᣨ���ᣩ FeCl3��Һ��Ũ��ˮ����ˮ����2�֣�
��2�֣�(5)ϡ���ᣨ���ᣩ FeCl3��Һ��Ũ��ˮ����ˮ����2�֣�
(6) ����2�֣���4��,���������𰸾����֣�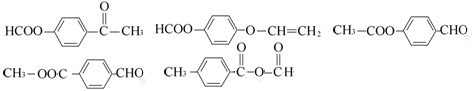
(7) ��Cl2��Br2 ��1�֣� FeCl3/Fe��������1�֣� ��ȡ����Ӧ��1�֣�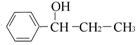 ��1�֣�
��1�֣�
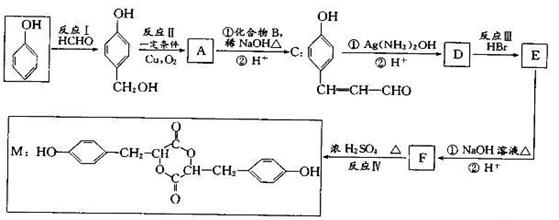
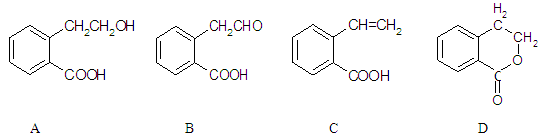
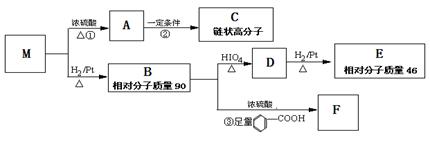
�������������2.8g�л���A����ȫȼ������0.15molCO2��1.8gH2O��ˮ�����ʵ�����0.1mol�����л�����Ԫ�ص�������2.8g��0.15mol��12g/mol��0.2mol��1g/mol��0.8g������ԭ�ӵ����ʵ�����0.8g��16g/mol��0.05mol�����л��������C��H��O����ԭ�ӵĸ���֮����0.15:0.2:0.05��3:4:1������A������ͼ��֪A����Է�������������60�����Է���ʽӦ����C3H4O��A�ܷ���������Ӧ��˵������ȩ������A�Ľṹ��ʽΪCH2��CHCHO��B�Ľṹ��ʽΪCH2��CHCOOH��B��״�����������Ӧ����C����C�Ľṹ��ʽΪCH2��CHCOOCH3��������֪��Ϣ������E�Ľṹ��ʽ��֪��D�Ľṹ��ʽΪ ��E������������Һ��ˮ�⣬�����ϼ�����F��F�к����ǻ����Ȼ������������ڵ�������Ӧ�γɻ���G��
��E������������Һ��ˮ�⣬�����ϼ�����F��F�к����ǻ����Ȼ������������ڵ�������Ӧ�γɻ���G��
��1���������Ϸ�����֪A�ķ���ʽΪC3H4O��
��2��C�к�������������Ϊ������F��G �ķ�Ӧ������ȡ����Ӧ��������Ӧ��
��3��A��������Һ��Ӧ�Ļ�ѧ����ʽΪCH2��CHCHO +2Ag(NH3)2OH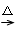 CH2��CHCOONH4 +2Ag��+ 3NH3 +H2O��
CH2��CHCOONH4 +2Ag��+ 3NH3 +H2O��
��4��D�Ľṹ��ʽΪ ��
��
��5�������㶹�أ� 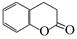 ��ˮ����з��ǻ����ɡ�����һ��ͬ���칹�壨
��ˮ����з��ǻ����ɡ�����һ��ͬ���칹�壨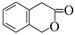 ��ˮ���õ����Ǵ��ǻ�����˼��������Ҫ�õ����Լ��У�NaOH��Һ��ϡ���ᣨ���ᣩ��FeCl3��Һ��Ũ��ˮ����ˮ����
��ˮ���õ����Ǵ��ǻ�����˼��������Ҫ�õ����Լ��У�NaOH��Һ��ϡ���ᣨ���ᣩ��FeCl3��Һ��Ũ��ˮ����ˮ����
��6����. �����г������⣬��������״�ṹ����.���������������ڶ�λ��ȡ��������. �ܷ���ˮ�ⷴӦ��������Na��Ӧ��˵�������������������ǻ�����.��������Cu(OH)2�����ʵ�����1:2����������Ӧ��˵��������1��ȩ����������������л���ͬ���칹��ṹ��ʽΪ ��
��
��7��������֪��Ϣ������ͼ��֪����Ӧ�����Ʊ�±��������Ӧ������Cl2��Br2��FeCl3/Fe��������M���ȱ�����Ӧ������ԭ�ӱ�ȡ������������ȡ����Ӧ����N�Ľṹ��ʽΪ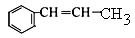 ��N�����е�̼̼˫����ˮ�����ӳɷ�Ӧ�����ǻ�����P�Ľṹ��ʽΪ
��N�����е�̼̼˫����ˮ�����ӳɷ�Ӧ�����ǻ�����P�Ľṹ��ʽΪ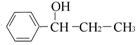 ��
��
���㣺�����л����ƶϡ������š��л���Ӧ���͡�ͬ���칹���жϡ�����ʽ��д�Լ���ϳ�·�����

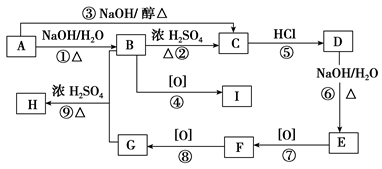
��ϩ��Ϊԭ�ϣ��ϳ�ijЩ�߾����·�����£�
��֪�� ����д��
����� ��
��
��1��CH3CH��CHCH3��������_______________��
��2��X�к��еĹ�������___________________��
��3��A��B�Ļ�ѧ����ʽ��__________________��
��4��D��E�ķ�Ӧ������_______________��
��5����Ϊ����F����NaHCO3��Ӧ����CO2��
�������й�˵����ȷ����_______________��
| A���л���Z�ܷ���������Ӧ | B���л���Y��HOCH2CH2OH��Ϊͬϵ�� |
| C���л���Y�ķе��B�� | D���л���F���뼺�������۳ɾۺ��� |
��Z��W�Ļ�ѧ����ʽ��__________________��
��6���߾���H�Ľṹ��ʽ��_________________��
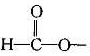
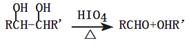
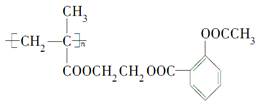

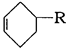
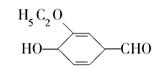 )��ʳƷ���Ӽ�������ԭ�ϣ�����ζ�����ȩ����Ũ����
)��ʳƷ���Ӽ�������ԭ�ϣ�����ζ�����ȩ����Ũ����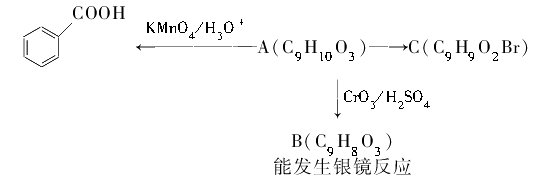 ��ʾ��
��ʾ�� b���뱽��ֱ��������̼ԭ��������ʱ����̼ԭ�Ӳſɱ�����KMnO4��Һ����Ϊ�Ȼ�
b���뱽��ֱ��������̼ԭ��������ʱ����̼ԭ�Ӳſɱ�����KMnO4��Һ����Ϊ�Ȼ�