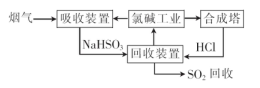
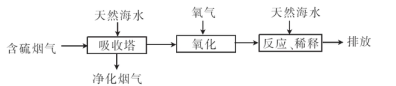
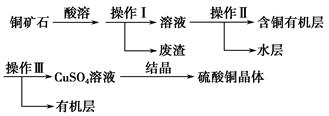
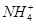��Ŀ����
ij������Һ���ܺ���Br����SO42-��H2SO3��NH4+�ȣ��ֱ�Ը���Һ��������ʵ�飺
�ټ���ʱ�ų��������ʹƷ����Һ��ɫ��
�ڼ������Һʹ��Һ�ʼ��ԣ��ټ���ʱ�ų��������ʹʪ��ĺ�ɫʯ����ֽ������
�ۼ�����ˮʱ����Һ�ԳʳȺ�ɫ���ټ���BaCl2��Һ������������ϡ����İ�ɫ������
�����������ʲ���ȷ������ԭ��Һ���Ƿ���ڵ��ǣ�������
�ټ���ʱ�ų��������ʹƷ����Һ��ɫ��
�ڼ������Һʹ��Һ�ʼ��ԣ��ټ���ʱ�ų��������ʹʪ��ĺ�ɫʯ����ֽ������
�ۼ�����ˮʱ����Һ�ԳʳȺ�ɫ���ټ���BaCl2��Һ������������ϡ����İ�ɫ������
�����������ʲ���ȷ������ԭ��Һ���Ƿ���ڵ��ǣ�������
| A��Br�� | B��SO42- | C��H2SO3 | D��NH4+ |
B
ʵ���ʹƷ����Һ��ɫ��������SO2���ʸ�ʵ���֤��ԭ��Һ��һ������H2SO3��ʵ���ʹʪ��ĺ�ɫʯ����ֽ������������NH3���ʿ�֤��ԭ��Һ��һ������NH4+��ʵ�����Һ�ԳʳȺ�ɫ������Cl2����Br�������嵥�ʵĽ�����ʸ�ʵ��֤��ԭ��Һ��һ������Br������ʵ�����ɵİ�ɫ������BaSO4������Ϊ��ˮ���������ԣ�����ȷ����ԭ��Һ�д���SO42-������H2SO3���������¡�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
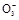 ������I
������I