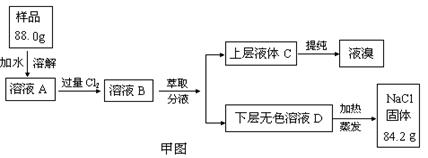
��Ŀ����
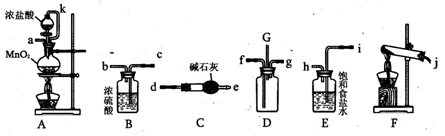
��16�֣�ij��ѧʵ��С���ͬѧΪ̽���ͱȽ�SO2����ˮ��Ư���ԣ���������µ�ʵ��װ�ã�ͼ3����

��1���ٷ�Ӧ��ʼһ��ʱ��۲쵽B��D�����Թ��е�Ʒ����Һ���ֵ������ǣ�
B��________________________________��D��____________________________��
��ֹͣͨ����,�ٸ�B��D�����Թֱܷ���ȣ������Թ��е�����ֱ�Ϊ
B��________________________________��D��____________________________��
��2����һ��ʵ��С���ͬѧ��ΪSO2����ˮ����Ư���ԣ�����Ϻ��Ư���Կ϶����ǿ�����ǽ��Ƶõ�SO2��Cl2��1:1ͬʱͨ�뵽Ʒ����Һ�У����������ɫЧ��������������������������������ԭ���û�ѧ����ʽ��ʾ��______________________________ ��
��3��װ��E����MnO2��Ũ���ᷴӦ�Ƶ�Cl2������Ӧ���ɵ�Cl2���Ϊ2.24L����״��������������HClΪ mol��
��4��ʵ���������ͬѧ��Ϊװ��C�п��ܺ���SO32����SO42����Cl����OH���������ӣ�����д��������SO42����SO32����ʵ�鱨�档
��ѡ�Լ���2 mol��L��1 HCl��1 mol��L��1 H2SO4��l mol��L��1 BaCl2��l mol��L��1MgCl2
1 mol��L��1 HNO3��0.1 mol��L��1 AgNO3�����Ʊ�����ˮ��

��1���ٷ�Ӧ��ʼһ��ʱ��۲쵽B��D�����Թ��е�Ʒ����Һ���ֵ������ǣ�
B��________________________________��D��____________________________��
��ֹͣͨ����,�ٸ�B��D�����Թֱܷ���ȣ������Թ��е�����ֱ�Ϊ
B��________________________________��D��____________________________��
��2����һ��ʵ��С���ͬѧ��ΪSO2����ˮ����Ư���ԣ�����Ϻ��Ư���Կ϶����ǿ�����ǽ��Ƶõ�SO2��Cl2��1:1ͬʱͨ�뵽Ʒ����Һ�У����������ɫЧ��������������������������������ԭ���û�ѧ����ʽ��ʾ��______________________________ ��
��3��װ��E����MnO2��Ũ���ᷴӦ�Ƶ�Cl2������Ӧ���ɵ�Cl2���Ϊ2.24L����״��������������HClΪ mol��
��4��ʵ���������ͬѧ��Ϊװ��C�п��ܺ���SO32����SO42����Cl����OH���������ӣ�����д��������SO42����SO32����ʵ�鱨�档
��ѡ�Լ���2 mol��L��1 HCl��1 mol��L��1 H2SO4��l mol��L��1 BaCl2��l mol��L��1MgCl2
1 mol��L��1 HNO3��0.1 mol��L��1 AgNO3�����Ʊ�����ˮ��
| ��� | ʵ����� | Ԥ������ͽ��� |
| ����� | ȡ��������Һ���Թ��У����� ������ | ��֤������Һ�к�SO32���� |
| ����� | �ڲ���ٵ���Һ�е������� | �� ֤������Һ�к�SO42���� |
���ڣ�4��С��ÿ��1�֣�����ÿ��2�֣���1����B��Ʒ����Һ��ɫ��D��Ʒ����Һ��ɫ��
��B����ɫ��Ʒ���ָֻ�Ϊ��ɫ��D������������
��2��Cl2��SO2��2H2O=2HCl��H2SO4 ��3��0.2
��4��
��B����ɫ��Ʒ���ָֻ�Ϊ��ɫ��D������������
��2��Cl2��SO2��2H2O=2HCl��H2SO4 ��3��0.2
��4��
| ��� | ʵ����� | Ԥ������ͽ��� |
| ����� | 2 mol��L-1HCl | �����ݲ��� |
| ����� | l mol��L-1BaCl2 | �а�ɫ�������� |
��1����SO2����Ư���ԣ���ʹƷ����Һ��ɫ����ˮ����ǿ�����ԣ�Ҳ��ʹƷ����Һ��ɫ��
������SO2��Ư�����ǿ���ģ����Լ�����ʹƷ����Һ�ָ���ɫ������ˮ��Ư�������������µģ��Dz�����ģ����Ȳ��ָܻ���ɫ��
��2������SO2�����л�ԭ�ԣ����������������ԣ����߷���������ԭ��Ӧ�����Ȼ�������ᣬ����ʽΪCl2��SO2��2H2O=2HCl��H2SO4��
��3��2.24L�����ڱ�״���µ����ʵ�����0.1mol��������ԭ���غ��֪�����������Ȼ�����0.2mol��
��4��SO32���ļ���������ᣬ��Ӧ������SO2���塣SO42���ļ������Ȼ�����Һ�����ɰ�ɫ�������ᱵ��
������SO2��Ư�����ǿ���ģ����Լ�����ʹƷ����Һ�ָ���ɫ������ˮ��Ư�������������µģ��Dz�����ģ����Ȳ��ָܻ���ɫ��
��2������SO2�����л�ԭ�ԣ����������������ԣ����߷���������ԭ��Ӧ�����Ȼ�������ᣬ����ʽΪCl2��SO2��2H2O=2HCl��H2SO4��
��3��2.24L�����ڱ�״���µ����ʵ�����0.1mol��������ԭ���غ��֪�����������Ȼ�����0.2mol��
��4��SO32���ļ���������ᣬ��Ӧ������SO2���塣SO42���ļ������Ȼ�����Һ�����ɰ�ɫ�������ᱵ��

��ϰ��ϵ�д�
 ������״Ԫ���Ծ�ϵ�д�
������״Ԫ���Ծ�ϵ�д�
�����Ŀ

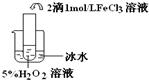
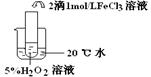
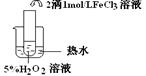

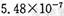 ��������������
��������������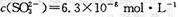 ��
��
 ��
��