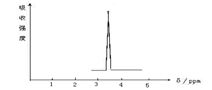
��Ŀ����
��ش����л�ѧ�й����⣺
��1���ִ���ҵ��ú�������ȿ������ȼ�ϵ������ʡ�����CO��SO2�ȵ��ŷţ��ֿ�������ˮú������;��
����֪��2C(s)+O2(g)��2CO(g)����H1 , 2H2(g)+O2(g)��2H2O(g)����H2 ��
��ӦC(s)+H2O(g) CO(g)+H2(g)����H�� �����ú���H1����H2�Ĵ���ʽ��ʾ��
CO(g)+H2(g)����H�� �����ú���H1����H2�Ĵ���ʽ��ʾ��
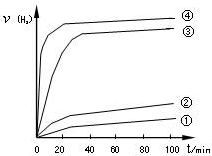
��CO��H2��һ�������ºϳɼ״��ķ�ӦΪ��CO(g)+2H2(g) CH3OH(g)����H3�������ݻ���Ϊ1L��a��b��c��d��e����ܱ������зֱ����1mol CO��2mol H2�Ļ�����壬���£�����ʵ���������������ͼ1��ͼ2��
CH3OH(g)����H3�������ݻ���Ϊ1L��a��b��c��d��e����ܱ������зֱ����1mol CO��2mol H2�Ļ�����壬���£�����ʵ���������������ͼ1��ͼ2��

a. �÷�Ӧ�Ħ�H3 0��ѡ���������������=������ͬ���� K1 K2��
b. ������d�е�ƽ��״̬ת�䵽����c�е�ƽ��״̬���ɲ�ȡ�Ĵ�ʩ�� ��
��2��ú�������ú���������ؽ�������ͼΪij�¶��£�Fe(OH)3(s)��Mg(OH)2(s)�ֱ�����Һ�дﵽ�����ܽ�ƽ��ı���Һ��pH������������Ũ�ȱ仯�������ͼ������
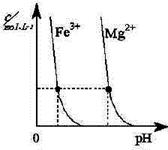
���¶��£��ܶȻ������Ĺ�ϵΪ��Ksp[ Fe(OH)3]____Ksp[Mg(OH)2]
���>��=��<��������������ɵ�Mg(OH)2��Һ�е���������Fe3+����ɫ������ȫ��ת��Ϊ���ɫ������ԭ����
��1���ִ���ҵ��ú�������ȿ������ȼ�ϵ������ʡ�����CO��SO2�ȵ��ŷţ��ֿ�������ˮú������;��
����֪��2C(s)+O2(g)��2CO(g)����H1 , 2H2(g)+O2(g)��2H2O(g)����H2 ��
��ӦC(s)+H2O(g)
 CO(g)+H2(g)����H�� �����ú���H1����H2�Ĵ���ʽ��ʾ��
CO(g)+H2(g)����H�� �����ú���H1����H2�Ĵ���ʽ��ʾ����CO��H2��һ�������ºϳɼ״��ķ�ӦΪ��CO(g)+2H2(g)
 CH3OH(g)����H3�������ݻ���Ϊ1L��a��b��c��d��e����ܱ������зֱ����1mol CO��2mol H2�Ļ�����壬���£�����ʵ���������������ͼ1��ͼ2��
CH3OH(g)����H3�������ݻ���Ϊ1L��a��b��c��d��e����ܱ������зֱ����1mol CO��2mol H2�Ļ�����壬���£�����ʵ���������������ͼ1��ͼ2��
a. �÷�Ӧ�Ħ�H3 0��ѡ���������������=������ͬ���� K1 K2��
b. ������d�е�ƽ��״̬ת�䵽����c�е�ƽ��״̬���ɲ�ȡ�Ĵ�ʩ�� ��
��2��ú�������ú���������ؽ�������ͼΪij�¶��£�Fe(OH)3(s)��Mg(OH)2(s)�ֱ�����Һ�дﵽ�����ܽ�ƽ��ı���Һ��pH������������Ũ�ȱ仯�������ͼ������

���¶��£��ܶȻ������Ĺ�ϵΪ��Ksp[ Fe(OH)3]____Ksp[Mg(OH)2]
���>��=��<��������������ɵ�Mg(OH)2��Һ�е���������Fe3+����ɫ������ȫ��ת��Ϊ���ɫ������ԭ����
��1����7�֣� �� (��H1����H2)/2 �� a.���� �� b. ���¡�������ӣ�ѹ
��2���� ����Ksp[ Fe(OH)3]��Ksp[Mg(OH)2] ��1�֣�����Һ�д����ܽ�ƽ�⣺Mg(OH)2 Mg2++2OH-��1�֣����¼����Fe3+��OH-���ɸ����ܽ��Fe(OH)3��ʹƽ����������ƶ���1�֣������Mg(OH)2ȫ���ܽ�ת��Ϊ����ɫ��Fe(OH)3��1�֣���
Mg2++2OH-��1�֣����¼����Fe3+��OH-���ɸ����ܽ��Fe(OH)3��ʹƽ����������ƶ���1�֣������Mg(OH)2ȫ���ܽ�ת��Ϊ����ɫ��Fe(OH)3��1�֣���
��2���� ����Ksp[ Fe(OH)3]��Ksp[Mg(OH)2] ��1�֣�����Һ�д����ܽ�ƽ�⣺Mg(OH)2
 Mg2++2OH-��1�֣����¼����Fe3+��OH-���ɸ����ܽ��Fe(OH)3��ʹƽ����������ƶ���1�֣������Mg(OH)2ȫ���ܽ�ת��Ϊ����ɫ��Fe(OH)3��1�֣���
Mg2++2OH-��1�֣����¼����Fe3+��OH-���ɸ����ܽ��Fe(OH)3��ʹƽ����������ƶ���1�֣������Mg(OH)2ȫ���ܽ�ת��Ϊ����ɫ��Fe(OH)3��1�֣�����1���ٸ��ݸ�˹���ɿ�֪�����٣��ڣ���2���õ�C(s)+H2O(g)  CO(g)+H2(g)�����Ԧ�H�� (��H1����H2)/2��
CO(g)+H2(g)�����Ԧ�H�� (��H1����H2)/2��
�ڸ���ͼ���֪���¶ȸߣ�ƽ��ʱ�״��ĵͣ�˵�������¶ȣ�ƽ�����淴Ӧ�����ƶ����������Ӧ�Ƿ��ȷ�Ӧ�������µ�ƽ�ⳣ����K1��K2��
��dת��Ϊc���״������ʵ������ӣ�˵��ƽ��������Ӧ�����ƶ�����˸��ݷ�Ӧʽ��֪����Ӧ�Ƿ��ȵġ������С�Ŀ��淴Ӧ�����Ըı�����������ǽ��»�������ӣ�ѹ��
��2������ͼ���֪���ڽ�������Ũ����ͬ�������£����������γɳ�����Ҫ��pHС�����������������ܶȻ�����С��������þ�ġ�����Ksp[ Fe(OH)3]��Ksp[Mg(OH)2]����Һ�д����ܽ�ƽ�⣺Mg(OH)2 Mg2++2OH-���¼����Fe3+��OH-���ɸ����ܽ��Fe(OH)3��ʹƽ����������ƶ������Mg(OH)2ȫ���ܽ�ת��Ϊ����ɫ��Fe(OH)3��
Mg2++2OH-���¼����Fe3+��OH-���ɸ����ܽ��Fe(OH)3��ʹƽ����������ƶ������Mg(OH)2ȫ���ܽ�ת��Ϊ����ɫ��Fe(OH)3��
 CO(g)+H2(g)�����Ԧ�H�� (��H1����H2)/2��
CO(g)+H2(g)�����Ԧ�H�� (��H1����H2)/2���ڸ���ͼ���֪���¶ȸߣ�ƽ��ʱ�״��ĵͣ�˵�������¶ȣ�ƽ�����淴Ӧ�����ƶ����������Ӧ�Ƿ��ȷ�Ӧ�������µ�ƽ�ⳣ����K1��K2��
��dת��Ϊc���״������ʵ������ӣ�˵��ƽ��������Ӧ�����ƶ�����˸��ݷ�Ӧʽ��֪����Ӧ�Ƿ��ȵġ������С�Ŀ��淴Ӧ�����Ըı�����������ǽ��»�������ӣ�ѹ��
��2������ͼ���֪���ڽ�������Ũ����ͬ�������£����������γɳ�����Ҫ��pHС�����������������ܶȻ�����С��������þ�ġ�����Ksp[ Fe(OH)3]��Ksp[Mg(OH)2]����Һ�д����ܽ�ƽ�⣺Mg(OH)2
 Mg2++2OH-���¼����Fe3+��OH-���ɸ����ܽ��Fe(OH)3��ʹƽ����������ƶ������Mg(OH)2ȫ���ܽ�ת��Ϊ����ɫ��Fe(OH)3��
Mg2++2OH-���¼����Fe3+��OH-���ɸ����ܽ��Fe(OH)3��ʹƽ����������ƶ������Mg(OH)2ȫ���ܽ�ת��Ϊ����ɫ��Fe(OH)3��
��ϰ��ϵ�д�
 ȫ�Ų��Ծ�ϵ�д�
ȫ�Ų��Ծ�ϵ�д� ���100��1�ž�ϵ�д�
���100��1�ž�ϵ�д�
�����Ŀ
 CH3OH(g) ��H< 0��
CH3OH(g) ��H< 0��
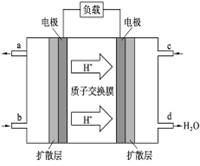
 N204(g)����ͬʱ�����N02���������ߣ���÷�Ӧ�ġ�H<O
N204(g)����ͬʱ�����N02���������ߣ���÷�Ӧ�ġ�H<O
 CO��g��+H2��g�����˷�Ӧ�����ȷ�Ӧ���ٴ˷�Ӧ�Ļ�ѧƽ�ⳣ������ʽΪ ��
CO��g��+H2��g�����˷�Ӧ�����ȷ�Ӧ���ٴ˷�Ӧ�Ļ�ѧƽ�ⳣ������ʽΪ �� CO (g)+2H2O (g) +519KJ����ҵ��Ҫѡ����ʵĴ������ֱ��X��Y��Z���ִ�����������ʵ�飨����������ͬ��
CO (g)+2H2O (g) +519KJ����ҵ��Ҫѡ����ʵĴ������ֱ��X��Y��Z���ִ�����������ʵ�飨����������ͬ�� N2(g)+2H2O(l) ��H="-a" kJ/mol
N2(g)+2H2O(l) ��H="-a" kJ/mol Cu2O+H2������������ӦʽΪ�� ��
Cu2O+H2������������ӦʽΪ�� ��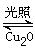 2H2(g)��O2(g) ��H����484 kJ��mol��1
2H2(g)��O2(g) ��H����484 kJ��mol��1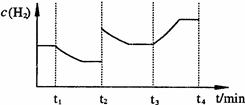
 ͼ��
ͼ��