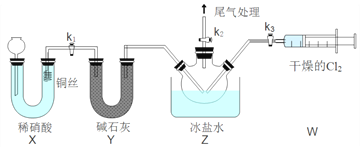
��Ŀ����
����Ŀ��ij��ѧѧϰС����������´Ӻ������պ�ĺ���������ȡ�ⵥ�ʵ����̣�

��1���ܽ⺣����ʱҪ�������2��3min��Ŀ����_________������a�������� _______��
��2�����ữ����ҺI�м���H2O2��Ŀ����__________________________________��
��3����֪I2��40%��NaOH��Һ��Ӧ���ɵ���������ͻ�ԭ��������ʵ���֮��Ϊ1:5��������Ӧ�Ļ�ѧ����ʽ��________________________________��
��4�������˵õ���I2��Ҫ����ϴ�Ӻ������ϴ�Ӽ�����Ӧ��ѡ�õ���______��
a.��ˮ b.�Ҵ� c.��ˮ d.����̼
��5����Na2S2O3�ı���Һ�ⶨ��Ʒ�Ĵ��ȣ�������Ӧ��I2+2Na2S2O3=Na2S4O6+2NaI��ȡ5.0g��Ʒ�����Ƴ�100ml��Һ��ȡ10.00ml��Һ���Ե�����ҺΪָʾ������Ũ��Ϊ0.050mol��L-1Na2S2O3�ı���Һ���еζ���������ݼ�¼���±���ʾ��
��� | 1 | 2 | 3 |
��Һ�����/mL | 10.00 | 10.00 | 10.00 |
����Na2S2O3����Һ�����/mL | 19.95 | 17.10 | 20.05 |
�ζ�ʱ���ﵽ�ζ��յ��������________���ⵥ���ڲ�Ʒ�е�����������________% ��
���𰸡� �ӿ�I-�ܽ⣬ʹ��������I-������ȫ���ܽ� ��Һ ��I-����ΪI2 3I2+6NaOH==NaIO3+5NaI+3H2O c �������һ�α���Һ����ɫ��Һǡ�ñ�Ϊ��ɫ���Ұ���Ӳ��ָ� 25.4
����������������������̿�֪�������Ҿ�ˮ������ˣ���Һ���ữ��˫��ˮ�ѵ�������������ȡ����Һ�����л����������������Һ���з���ȡ����Һ��������Һ�����ữ�����˵õ��⡣
��1���ܽ⺣����ʱҪ�������2��3min��Ŀ���Ǽӿ�I-�ܽ⣬ʹ��������I-������ȫ���ܽ⣬����a�������Ƿ�Һ��
��2�����ữ����ҺI�м���H2O2��Ŀ���ǽ�I-����ΪI2��
��3��I2��40%��NaOH��Һ��Ӧ���ɵ���������ͻ�ԭ��������ʵ���֮��Ϊ1:5����ⷢ���绯��Ӧ����Ԫ�صĻ��ϼ۲��ֽ�Ϊ-1�ۡ��������ߵ�+5�ۣ��ʷ�����Ӧ�Ļ�ѧ����ʽ��3I2+6NaOH==NaIO3+5NaI+3H2O��
��4�������˵õ���I2��Ҫ����ϴ�Ӻ��Ϊ���ٵ���ܽ���ʧ��Ӧѡ����ˮ����ϴ�Ӽ�����Ӧ��ѡ�õ���c��
��5���ζ�ʱ���ﵽ�ζ��յ�������Ǽ������һ�α���Һ����ɫ��Һǡ�ñ�Ϊ��ɫ���Ұ���Ӳ��ָ����ɱ������ݿ�֪����2��ʵ�����������������Ӧ��ȥ�����ݣ��ɵ�1�κ͵�3�ε�������ƽ��ֵ���õ�����Na2S2O3����Һ�����Ϊ20.00mL����I2+2Na2S2O3=Na2S4O6+2NaI��֪��n(I2)=0.5n(Na2S2O3)=![]() �����ԣ��ⵥ���ڲ�Ʒ�е�����������
�����ԣ��ⵥ���ڲ�Ʒ�е�����������![]() 25.4% ��
25.4% ��

 ����ȫ���ִʾ��ƪ��ϵ�д�
����ȫ���ִʾ��ƪ��ϵ�д� �����߿����ϵ�д�
�����߿����ϵ�д�