��Ŀ����
��1��������������������___________________��
��2���кͷ�Ӧ�ų���������������������ڷ�Ӧ�лӷ������õ��к��ȵ���ֵ________���ƫ����ƫС�����䡱����
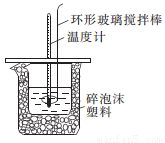
��3����ʵ��С����������ʵ�飬ÿ��ȡ��Һ��50 mL������ʵ�����ݼ�¼���£�
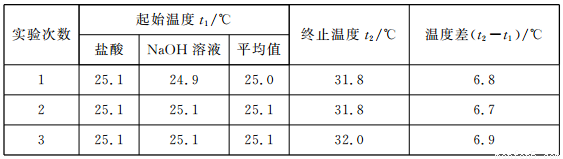
��֪���ᡢNaOH��Һ���ܶ���ˮ��ͬ���кͺ����ɵ���Һ�ı�����c=4��18��10-3kJ��(g����)����÷�Ӧ���к��ȡ�H=__________��
��4�����к��Ȳⶨʵ���У�������ʼ�¶�ʱ��Ҫ�����¶ȼƣ����ֻ��һ���¶ȼƲ��������õ��к��ȵ���ֵ___________���ƫ����ƫС�����䡱����
��1����һѧ����ʵ���Ҳ�ij��ҺpH��ʵ��ʱ������������ˮ��ʪpH��ֽ��Ȼ���ýྻ����IJ�����պȡ�������м�⡣
�����ִ������ ���һ����/��һ������/����һ�������ᵼ��ʵ��������
�������˷��ֱ�ⶨc(H+)��ȵ�����ʹ�����Һ��pH�����ϴ���� ��
��2��������֪Ũ�ȵ� NaOH ��Һ�ⶨij HCl��Һ��Ũ�ȣ��ο���ͼ���ӱ���ѡ����ȷ���
��� | ��ƿ����Һ | �ζ�������Һ | ѡ��ָʾ�� |
|
A | �� | �� | ʯ | ���ң� |
B | �� | �� | ��̪ | ���ף� |
C | �� | �� | ���� | ���ף� |
D | �� | �� | ��̪ | ���ң� |
(3)���ñ���NaOH�ζ�δ֪Ũ�ȵ����ᣬѡ�÷�̪Ϊָʾ������ɲⶨ���ƫ�ߵ�ԭ������� ��
A�����Ʊ���Һ�����������л���Na2CO3����
B���ζ��յ����ʱ�����ӵζ��ܵĿ̶ȣ�������������ȷ
C��ʢװδ֪Һ����ƿ������ˮϴ����δ�ô���Һ��ϴ
D���ζ����յ����ʱ���ֵζ��ܼ��촦����һ����Һ
E��δ�ñ�Һ��ϴ��ʽ�ζ���
�����±�(���ֶ�����Ԫ�ص�ԭ�Ӱ뾶����Ҫ���ϼ�)��Ϣ�������жϲ���ȷ����
Ԫ�ش��� | A | B | C | D | E |
ԭ�Ӱ뾶/nm | 0.186 | 0.143 | 0.089 | 0.102 | 0.074 |
��Ҫ���ϼ� | +1 | +3 | +2 | +6��-2 | -2 |
A�������ԣ�A��C B���⻯��ķе�H2D��H2E
C����һ�����ܣ� A>B D�����ʵ��۵㣺A<B


 ѡ�õζ���
ѡ�õζ��� о
о 2AB3(g)�Ļ�ѧƽ��״̬��Ӱ��������ͼ��ʾ��T��ʾ�¶ȣ�n��ʾ���ʵ������������ж���ȷ���ǣ� ��
2AB3(g)�Ļ�ѧƽ��״̬��Ӱ��������ͼ��ʾ��T��ʾ�¶ȣ�n��ʾ���ʵ������������ж���ȷ���ǣ� ��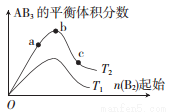
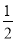 H2SO4��Ũ��=
H2SO4��Ũ��= Na2SO4(aq)+H2O(1) ��H3
Na2SO4(aq)+H2O(1) ��H3