��Ŀ����
8����֪ͭԪ�����γɶ��ֻ����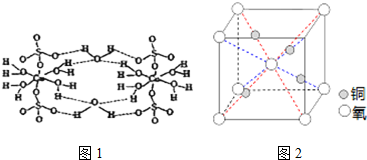
��1��д����̬Cu��M�ܲ�ĵ����Ų�ʽ3s23p63d10��Cuͬ���ڵ�Ԫ���У���ͭԭ��������������ȵ�Ԫ�ػ���K��Cr��Ga����Ԫ�ط��ţ�
��2��CuSO4•5H2OҲ��д��[Cu��H2O��4]SO4•H2O����ṹʾ��ͼ��ͼ1��ʾ��
�þ���������Ԫ�ص縺���ɴ�С˳����O��S��H��Cu���þ����������Ӿ��壬�þ�������ˮ���Ӳ����γɵ����������������λ����
��3��Cu2+����NH3��Cl-���γ���λ��Ϊ4��������֪[Cu��NH3��4]2+���жԳƵĿռ乹�ͣ�[Cu��NH3��4]2+�е�����NH3������Cl-ȡ�����ܵõ����ֲ�ͬ�ṹ�IJ����[Cu��NH3��4]2+�Ŀռ乹��Ϊƽ���ı��Σ�NH3��VSEPR ģ��Ϊ������
��4��ͭ�ǵ�����������Ҫ�Ĺ���Ԫ��֮һ���䵥�ʼ���������й㷺��;��ͭ������ͭԭ�Ӷѻ�ģ��Ϊ�����������ܶѻ���ͭ��ij�������ᄃ���ṹ��ͼ2��ʾ�����þ�����ܶ�Ϊd g/cm3�������ӵ�������ֵΪNA����þ�����ͭԭ������ԭ��֮����̵ľ���Ϊ$\frac{\sqrt{3}}{4}$��$\root{3}{\frac{288}{a{N}_{A}}}$cm�����ú�d��NA��ʽ�ӱ�ʾ�����ػ���
���� ��1��Cuԭ�Ӻ�����29�����ӣ���������Ų�Ϊ1s22s22p63s23p63d104s1���ݴ˿���дM�ܲ�ĵ����Ų�ʽ��CuԪ��λ�����ڱ��������ڣ�Cuԭ���������1�����ӣ����Ԫ�����ڱ����ҳ�������������ͭԭ��������������ȵ�Ԫ�أ�
��2��CuSO4•5H2O�����к�Cu��S��O��HԪ�أ�����Ԫ�صķǽ�����Խǿ���縺��Խ����жϵ縺�Դ�С���þ����к�ͭ���ӡ���������ӣ��������Ӿ��壬����ͼ1�ṹ��֪ˮ���Ӳ����γɵ�������Ϊ�������λ����
��3��Cu2+����NH3��Cl-���γ���λ��Ϊ4��[Cu��NH3��4]2+�е�����NH3������Cl-ȡ�����ܵõ����ֲ�ͬ�ṹ�IJ��[Cu��NH3��4]2+�Ŀռ乹��ӦΪƽ���ı��νṹ������NH3�����е�ԭ�ӵļ۲���Ӷ����ж�VSEPR ģ�ͣ�
��4��ͭ������ͭԭ�Ӷѻ�ģ��Ϊ�����������ܶѻ����ɾ�̯������������ͭ������Cuԭ�Ӻ�Oԭ�ӵ���Ŀ�������ܶȼ��㹫ʽ$��=\frac{m}{V}$�����������ټ���ͭԭ������ԭ��֮��ľ��뼴�ɣ�
��� �⣺��1��Cuԭ�Ӻ�����29�����ӣ���������Ų�Ϊ1s22s22p63s23p63d104s1��M�ܲ�ĵ����Ų�ʽΪ3s23p63d10��CuԪ��λ�����ڱ��������ڣ�Cuԭ���������1�����ӣ�������������ͭԭ��������������ȵ�Ԫ����K��Cr��Ga��
�ʴ�Ϊ��3s23p63d10��K��Cr��Ga��
��2��CuSO4•5H2O�����к�Cu��S��O��HԪ�أ�����Ԫ�صķǽ�����Խǿ���縺��Խ���֪�縺���ɴ�С˳����O��S��H��Cu���þ����к�ͭ���ӡ���������ӣ��������Ӿ��壬����ͼ1�ṹ��֪ˮ���Ӳ����γɵ�������Ϊ�������λ����
�ʴ�Ϊ��O��S��H��Cu�����ӣ��������λ����
��3��Cu2+����NH3��Cl-���γ���λ��Ϊ4��[Cu��NH3��4]2+�е�����NH3������Cl-ȡ�����ܵõ����ֲ�ͬ�ṹ�IJ��[Cu��NH3��4]2+�Ŀռ乹��ӦΪƽ���ı��νṹ������NH3�����е�ԭ�ӵļ۲���Ӷ���Ϊ$\frac{5+3}{2}$=4������NH3��VSEPRģ��������ṹ��
�ʴ�Ϊ��ƽ���ı��Σ������壻
��4��ͭ������ͭԭ�Ӷѻ�ģ��Ϊ�����������ܶѻ�����ͭ��ij�������ᄃ���У�Oԭ���ھ����Ķ�������ģ���Oԭ����=$\frac{1}{8}$��8+1=2��Cuԭ��ȫ�������ģ���Cuԭ����=4����һ��������ͭ��������2��Oԭ�Ӻ�4��Cuԭ�ӣ������ܶȼ��㹫ʽ$��=\frac{m}{V}$��֪�����V=$\frac{m}{��}$=$\frac{\frac{288}{{N}_{A}}}{a}$cm3�����Ծ����ı߳�Ϊ$\root{3}{\frac{288}{a{N}_{A}}}$cm�����ݾ����Ľṹͼ��֪��������ͭԭ������ԭ��֮��ľ��뾧���߳���$\frac{\sqrt{3}}{4}$�����Ըþ�����ͭԭ������ԭ��֮��ľ���Ϊ$\frac{\sqrt{3}}{4}$��$\root{3}{\frac{288}{a{N}_{A}}}$cm��
�ʴ�Ϊ�������������ܶѻ���$\frac{\sqrt{3}}{4}$��$\root{3}{\frac{288}{a{N}_{A}}}$��
���� ������Ҫ�����˾����ļ��㡢��������Ų����ӻ������ʽ�Լ����ӹ��ͺ����������ݣ��ۺ��Խ�ǿ���Ѷ��еȣ����þ�̯�����㾧������ɣ�

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�| A�� | �ƾ���Ũ����������Ϊ3��1�����������м���ƾ�������������������Ũ���ᣬ�ӱ���ȴ�߽��� | |
| B�� | �¶ȼƵ�ˮ����Ӧ���뷴Ӧ���Һ��Һ���£����Ʒ�Ӧ�¶�170�� | |
| C�� | Ӧ�ڻ��Һ�мӼ�Ƭ���Ƭ��ֹ���� | |
| D�� | ŨH2SO4�����ã���������ˮ�� |
| A�� | 1��1��1 | B�� | 1��2��3 | C�� | 1��4��2 | D�� | 3��2��1 |
| A�� | ��Al2��SO4��3��Һ�е��������ˮ��Al3++4NH3•H2O=AlO${\;}_{2}^{-}$+4NH${\;}_{4}^{+}$+2H2O | |
| B�� | ��NaOH��Һ��ͨ��Cl2��2OH-+Cl2=Cl-+ClO-+H2O | |
| C�� | ��CaCO3�м���ϡ���CaCO3+2H+=Ca2++CO2��+H2O | |
| D�� | ��FeBr2��Һ��ͨ��������Cl2��2Fe2++Cl2=2Fe3++2Cl- |
| A�� | ���Ľṹ��P4��ͬ | |
| B�� | ����Ħ������Ϊ64 | |
| C�� | ����������ͬ�������� | |
| D�� | ����������������ͨ�����Ӽ���������϶��ɵ����� |
| A�� | SiO2 | B�� | CCl4 | C�� | NaCl | D�� | H2O |