��Ŀ����
��1������������ȼ�ϵ�صĹ���ԭ����ͼ��ʾ����֪����е����Ϊ���ڹ��������O2�����������������ƶ�����ȼ����ΪC2H4ʱ����������������Ӧʽ�ֱ�Ϊ
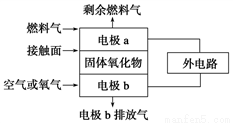
������______________________��
������____________________��
��2����ⷨ�������״���ˮ����ɵ���Ⱦ��ԭ���ǣ�ͨ�罫Co2��������Co3����Ȼ��Co3�����״�������CO2��H��(��ʯīϩ������ȥCo2��)��������ͼ��ʾװ��ģ���������̣���Co2���������ĵ缫��ӦʽΪ__________________����ȥ�״������ӷ���ʽΪ___________��
��3����ͼΪ��ͭ���ڳ�ʪ�����з����ĵ绯ѧ��ʴ��ʾ��ͼ��
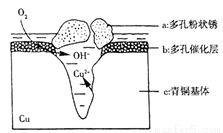
�ٻ����е�Cl����ɢ���ڣ�����������Ӧ���������Ӧ�����������ɶ��״��Cu2(OH)3Cl�������ӷ���ʽΪ______________________��
��������4.29 g Cu2(OH)3Cl���������Ϻ��������Ϊ___________L����״������
��ϰ��ϵ�д�
 һ����ʦ�����Ծ�ϵ�д�
һ����ʦ�����Ծ�ϵ�д�
�����Ŀ





 ]
] 2AB3��g������H<0������ͼ����ȷ���ǣ�
2AB3��g������H<0������ͼ����ȷ���ǣ�
 O2(g)��2CO2(g) ��3H2O(l)��H =��1560kJ��mol��1
O2(g)��2CO2(g) ��3H2O(l)��H =��1560kJ��mol��1 O2(g)��2CO2(g) ��3H2O(l)��H =��52.0kJ��mol��1
O2(g)��2CO2(g) ��3H2O(l)��H =��52.0kJ��mol��1