��Ŀ����
X��Y��Z��M��WΪ���ֶ�����Ԫ�ء�X��Y��Z��ԭ���������ε�����ͬ����Ԫ�أ�������������֮��Ϊ15��X��Z���γ�XZ2���ӣ�Y��M�γɵ���̬�������ڱ�״���µ��ܶ�Ϊ0.76 g/L��W����������X��Y��Z��M����Ԫ��������֮�͵�1/2������˵���������
A. ԭ�Ӱ뾶��W��X��Y��Z��M
B. XZ2Ϊֱ���εĹ��ۻ�����
C. X��Y��Z �ֱ���MԪ���γɵ��������ķе���������
D. ��X��Y��Z��M����Ԫ���γɵĻ�����һ���������Ӽ����ۼ�
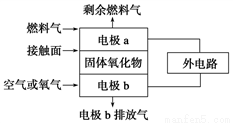
����ͼ��ʾװ�ý�������ʵ�飬a��b��c����װ�Լ����±���ʾ������ʵ����������۶�Ӧ��ϵ��ȷ��һ����

ѡ�� | a | b | c | ���� | ���� |
A | ����ʳ��ˮ | ̼���� | ��ˮ | c����Һ��ɫ��ȥ | ��Ȳ������ԭ��Ӧ |
B | Ũ���� | KMnO4���� | NaBr��Һ | c����Һ����ɫ���ɫ | Cl2�������Ա�Br2ǿ |
C | ϡ���� | ����ʯ | Na2SiO3��Һ | c���а�ɫ��״�������� | ̼������Աȹ���ǿ |
D | ���� | Na2SO3���� | ����KMnO4��Һ | c����Һ��ɫ��ȥ | SO2����Ư���� |
A. A B. B C. C D. D
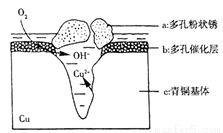
��ҵ����ú��ˮΪԭ��ͨ��һϵ��ת���ɱ�Ϊ�����Դ������ҵԭ�ϼ״���
��1����֪��C(s)+O2(g)=CO2(g) ��H1
��2H2(g)+O2(g)=2H2O (l) ��H2
��H2O (l)= H2O (g) ��H3
��̼��ˮ������ӦC(s)+2H2O(g) CO2(g)+2H2(g)�Ħ�H =________��
CO2(g)+2H2(g)�Ħ�H =________��
��2����ҵ��Ҳ���Խ�����������Ӧ�õ���CO2��H2��һ���ϳɼ״�����Ӧ����ʽΪ��CO2(g)��3H2(g) CH3OH(g)��H2O(g����H��0
CH3OH(g)��H2O(g����H��0
�ٹ�ҵ����������CO2��H2��ת����________���ǰ�ߴ������ߴ���һ�������жϡ�����Ϊ����״��IJ��ʿ��Բ�ȡ�Ĵ�ʩ��_______________�������㣩��
����һ���º����ܱ������г���1 mol CO2��3 mol H2����������Ӧ�����CO2��CH3OH(g)Ũ����ʱ��仯����ͼ��ʾ�����¶��µ�ƽ�ⳣ��Ϊ______��������λ��Ч���֣���
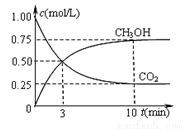
�ı��¶ȣ�ʹ��ӦCO2(g)+3H2(g) CH3OH(g)+H2O(g)�е��������ʶ�Ϊ��̬����ʼ�¶������ͬ��T1�桢2 L�ܱ�����������Ӧ�����в������ݼ��±���
CH3OH(g)+H2O(g)�е��������ʶ�Ϊ��̬����ʼ�¶������ͬ��T1�桢2 L�ܱ�����������Ӧ�����в������ݼ��±���
��Ӧʱ�� | CO2��mol�� | H2��mol�� | CH3OH��mol�� | H2O��mol�� | |
��Ӧ�� ���º��� | 0min | 2 | 6 | 0 | 0 |
10min | 4.5 | ||||
20min | 1 | ||||
30min | 1 | ||||
��Ӧ�� ���Ⱥ��� | 0min | 0 | 0 | 2 | 2 |
�ٴﵽƽ��ʱ����Ӧ��Աȣ�ƽ�ⳣ��K(��)___K(��)�����������������=������ͬ����ƽ��ʱCH3OH��Ũ��c(��)___c(��)��
�ڶԷ�Ӧ��ǰ10 min�ڵ�ƽ����Ӧ���ʦ�(CH3OH)=______����30 minʱֻ���������ٳ���1 mol CO2(g)��1 mol H2O(g)����ƽ��_____�ƶ��������������������
��һ������H2O(g)��CO(g)�ֱ�ͨ�뵽һ���Ϊ2 L�ĺ����ܱ������н��з�Ӧ��H2O(g)��CO(g)CO2(g)��H2(g)���õ������������ݣ��ݴ˻ش��������⣺
ʵ���� | �¶� | ��ʼ�� | ƽ���� | �ﵽƽ������ʱ�� | ||
H2O | CO | H2 | CO | |||
1 | 650�� | 1 mol | 2 mol | 0.8 mol | 1.2 mol | 5 min |
2 | 900�� | 0.5 mol | 1 mol | 0.2 mol | 0.8 mol | 3 min |
3 | 900�� | a | b | c | d | t |
��1�������������ݣ�ʵ��1����v(CO2)��ʾ��Ӧ����Ϊ_______���Ԧ�(CO)��ʾת����Ϊ_____��
�ڸ÷�Ӧ��650��ʱƽ�ⳣ����ֵΪ_________���÷�Ӧ���淴ӦΪ_________ (������š�)�ȷ�Ӧ��
����ʵ��3��ʵ��2��CO�İٷֺ�����ͬ����t<3 min����a��bӦ����Ĺ�ϵ��_______��
��2����ͼ1��2��ʾ������Ӧ��ʱ��t1�ﵽƽ�⣬��ʱ��t2��ı�ij�������������仯�������
��ͼ1��ʱ��t2�����ı������������____________(дһ�����ɣ���ͬ)��
��ͼ2��ʱ��t2�����ı������������______________________________________��

��3����850 ��ʱ����ƽ�ⳣ��K��1��850 ��ʱ�ڸ�������ͬʱ����1.0 mol CO��3.0 mol H2O��1.0 mol CO2��5.0 mol H2����ʱ��Ӧ��________(�����Ӧ�����淴Ӧ��)������У�ƽ��ʱCO2�����ʵ���Ϊ__________��