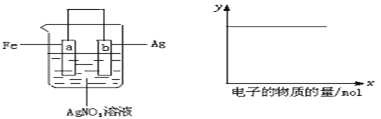
��Ŀ����
����Ŀ����������(ClNO)���л��ϳ��е���Ҫ�Լ�����ͨ�����·�Ӧ��ã�2NO(g)��Cl2(g) ![]() 2ClNO(g)��
2ClNO(g)��
��1�����������������ڴ����еĺ������������ʱ�������������ȣ��漰���·�Ӧ��
��2NO2(g)��NaCl(s) ![]() NaNO3(s)��ClNO(g)��K1
NaNO3(s)��ClNO(g)��K1
��4NO2(g)��2NaCl(s) ![]() 2NaNO3(s)��2NO(g)��Cl2(g)��K2
2NaNO3(s)��2NO(g)��Cl2(g)��K2
��2NO(g)��Cl2(g) ![]() 2ClNO(g)��K3
2ClNO(g)��K3
��K3��________(��K1��K2��ʾ)��
��2����֪���ֻ�ѧ���ļ����������±�(�������ȵĽṹΪCl��N===O)��
��ѧ�� | N��O | Cl��Cl | Cl��N | N===O |
����/(kJ��mol��1) | 630 | 243 | a | 607 |
��2NO(g)��Cl2(g) ![]() 2ClNO(g)��Ӧ�Ħ�H��a�Ĺ�ϵΪ��H��_______kJ��mol��1��
2ClNO(g)��Ӧ�Ħ�H��a�Ĺ�ϵΪ��H��_______kJ��mol��1��
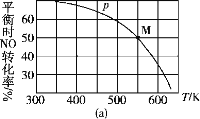
��3����Ͷ�ϱ�2��1��NO��Cl2���뵽һ��ѹ���ܱ������з�����Ӧ��ƽ��ʱNO��ת�������¶�T��ѹǿp(��ѹ)�Ĺ�ϵ��ͼ(a)��ʾ��
�ٸ÷�Ӧ�Ħ�H________(�>����<������)0��
����pѹǿ�����£�M��ʱ������NO���������Ϊ________��
������Ӧһֱ������pѹǿ�����½��У���M��ķ�ѹƽ�ⳣ��Kp��________(�ú�p�ı���ʽ��ʾ����ƽ���ѹ����ƽ��Ũ�ȼ��㣬��ѹ����ѹ���������)��
 ��
��
��4��һ���������ں��º��ݵ��ܱ������а�һ����������NO(g)��Cl2(g)����n(NO)/n(Cl2)��1.5ʱ�ﵽƽ��״̬��ClNO���������������ͼ(b)��D��E��F�����е�________�㡣
���𰸡�K3=K12/K2 +(289-2a) < 40% 5/p D
��������
��1����֪����2NO2��g��+NaCl��s��![]() NaNO3��s��+ClNO��g������4NO2��g��+2NaCl��s��
NaNO3��s��+ClNO��g������4NO2��g��+2NaCl��s��![]() 2NaNO3��s��+2NO��g��+Cl2��g�������١�2-�ڿɵã�2NO��g��+Cl2��g��
2NaNO3��s��+2NO��g��+Cl2��g�������١�2-�ڿɵã�2NO��g��+Cl2��g��![]() 2ClNO��g������ƽ�ⳣ��K3=K12/K2���ʴ�Ϊ��K3=K12/K2��
2ClNO��g������ƽ�ⳣ��K3=K12/K2���ʴ�Ϊ��K3=K12/K2��
��2��2NO��g��+C12��g��![]() 2C1NO��g����Ӧ�ġ�H=��Ӧ��ļ���֮��-������ļ���֮��=��2��630+243��-��2a+2��607��=��289-2a��kJ/mol���ʴ�Ϊ��289-2a��
2C1NO��g����Ӧ�ġ�H=��Ӧ��ļ���֮��-������ļ���֮��=��2��630+243��-��2a+2��607��=��289-2a��kJ/mol���ʴ�Ϊ��289-2a��
��3���ٸ���ͼ���֪T1<T2�������¶�c��C1NO������˵��ƽ��������Ӧ�����ƶ���˵������Ӧ�Ƿ��ȷ�Ӧ����H<0���ʴ�Ϊ��<��
������ʼn(NO)Ϊ2mol��n(Cl2)Ϊ1mol����ͼ��֪NOת����Ϊ50%�������⽨����������ʽ��
2NO��g��+Cl2��g��![]() 2ClNO��g��
2ClNO��g��
��ʼ��mol�� 2 1 0
��Ӧ��mol�� 1 0.5 1
ƽ�⣨mol�� 1 0.5 1
ƽ��ʱ�������������ʵ���Ϊ2.5mol����M��ʱ������NO���������Ϊ1mol/2.5mol��100%=40%,�ʴ�Ϊ��40%��
��������ʽ���ݼ���ɵã�NO���������Ϊ40%��Cl2���������Ϊ20%��ClNO���������Ϊ40%����M��ķ�ѹƽ�ⳣ��Kp����0.4p��2/��0.4p��2����0.2p��=5/p���ʴ�Ϊ��5/p��
��4��n(NO)/n(Cl2)�ı�ֵԽС��˵����n��NO�����䣬n��C12��Խ������NO��ת����Խ��NO��ת����������A�㣻����Ӧ������ʵ���֮�ȵ����������֮��ʱ�����ַ�Ӧ���ת������ͬ���ʴ�ʱ������İٷֺ������n(NO)/n(Cl2) ����1.5ʱ����İٷֺ�����������Ӧ����D�㣬�ʴ�Ϊ��D��

����Ŀ��һ���¶��£���1 L�����ܱ������м���1 mol��N2(g)��3 mol H2(g)������Ӧ��N2(g)��3H2(g)![]() 2NH3(g)����H<0��NH3�����ʵ�����ʱ��Ĺ�ϵ�����ʾ������˵���������(����)
2NH3(g)����H<0��NH3�����ʵ�����ʱ��Ĺ�ϵ�����ʾ������˵���������(����)
ʱ��/min | 0 | t1 | t2 | t3 |
NH3���ʵ���/mol | 0 | 0.2 | 0.3 | 0.3 |
A. 0��t1 min��v(NH3)��0.2/t1 mol��L��1��min��1
B. �����¶ȣ���ʹ����Ӧ���ʼ�С���淴Ӧ��������ƽ�������ƶ�
C. N2(g)��3H2(g)![]() 2NH3(g)�Ļ��С��2NH3(g)
2NH3(g)�Ļ��С��2NH3(g)![]() N2(g)��3H2(g)�Ļ��
N2(g)��3H2(g)�Ļ��
D. t3 minʱ�ټ���1 mol��N2(g)��3 mol H2(g)����Ӧ�ﵽ��ƽ��ʱ��c(N2)>0.85 mol��L��1