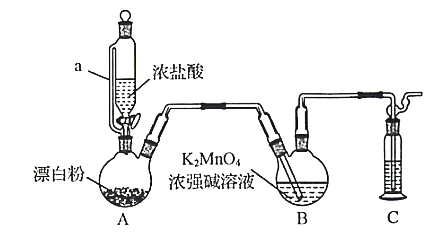
ЬтФПФкШн
ЁОЬтФПЁПЛЦЭПѓЃЈжївЊГЩЗжЮЊЖўСђЛЏбЧЬњЭЃКCuFeS2ЃЉЪЧжЦШЁЭМАЦфЛЏКЯЮяЕФжївЊдСЯжЎвЛЃЌЛЙПЩжЦБИСђМАЬњЕФЛЏКЯЮяЁЃвБСЖЭЕФЗДгІЮЊ8CuFeS2+21O2 ![]() 8Cu+4FeO+2Fe2O3+16SO2 ЁЃ
8Cu+4FeO+2Fe2O3+16SO2 ЁЃ
ЃЈ1ЃЉШєCuFeS2жаFeЕФЛЏКЯМлЮЊ+2ЃЌЗДгІжаБЛЛЙдЕФдЊЫиЪЧ__________ЃЈЬюдЊЫиЗћКХЃЉЁЃ
ЃЈ2ЃЉгУЯЁH2SO4НўХнЩЯЪіЗДгІжЦШЁЭКѓЪЃгрЕФЙЬЬхВадќЃЌШЁЩйСПЫљЕУШмвКЃЌМьбщШмвКжаДцдкFe3ЃЋЕФЗНЗЈЪЧ_________ЃЈзЂУїЪдМСЁЂЯжЯѓЁЂНсТлЃЉЁЃ
ЃЈ3ЃЉЩЯЪівБСЖЙ§ГЬВњЩњДѓСПSO2ЁЃЯТСаДІРэЗНАИжаКЯРэЕФЪЧ_____________ЃЈЬюДњКХЃЉ
a ИпПеХХЗХ b гУгкжЦБИСђЫс c гУДПМюШмвКЮќЪежЦNa2SO3 d гУХЈСђЫсЮќЪе
ЃЈ4ЃЉбщжЄЛЦЭПѓвБСЖЭЕФЗДгІКѓЦјЬхжаКЌгаSO2ЕФЗНЗЈЪЧ________________ЁЃ
ЃЈ5ЃЉЪЕбщЪвжЦБИЃЌЪеМЏИЩдяЕФSO2ЃЌЫљашвЧЦїШчЯТЁЃ

ЂйЦфжазАжУAВњЩњSO2 ЃЌЗДгІЕФЛЏбЇЗНГЬЪНЮЊ_______________ЁЃ
ЂкЧыАДЦјСїЗНЯђСЌНгИївЧЦїНгПкЃЌЫГађЮЊa Ёњ_____Ёњ_______Ёњ________Ёњ _______Ёњ f ЃЌзАжУEжаNaOHШмвКЕФзїгУЪЧ____________ЁЃ
ЁОД№АИЁПCuЁЂO ШЁЩйСПШмвКЃЌЕЮМгKSCNШмвКЃЌШмвКБфКь bc НЋЦјЬхЭЈШыЦЗКьШмвКжаЃЌШчЙћЦЗКьШмвКЭЪЩЋЃЌМгШШКѓгжБфКьЃЌдђжЄУїгаSO2 Cu+2H2SO4ЃЈХЈЃЉ![]() CuSO4+SO2Ёќ+2H2O d e c b ЮќЪеЖргрЕФSO2ЃЌЗРжЙЮлШОПеЦј
CuSO4+SO2Ёќ+2H2O d e c b ЮќЪеЖргрЕФSO2ЃЌЗРжЙЮлШОПеЦј
ЁОНтЮіЁП
(1)8CuFeS2+21O2 ![]() 8Cu+4FeO+2Fe2O3+16SO2жаЃЌCuдЊЫиЕФЛЏКЯМлгЩ+2МлНЕЕЭЮЊ0ЁЂOдЊЫиЕФЛЏКЯМлгЩ0НЕЕЭЮЊ-2МлЃЌFeдЊЫиЕФЛЏКЯМлгЩ+2МлЩ§ИпЮЊ+3МлЁЂSдЊЫиЕФЛЏКЯМлгЩ-2МлЩ§ИпЮЊ+4МлЃЛ
8Cu+4FeO+2Fe2O3+16SO2жаЃЌCuдЊЫиЕФЛЏКЯМлгЩ+2МлНЕЕЭЮЊ0ЁЂOдЊЫиЕФЛЏКЯМлгЩ0НЕЕЭЮЊ-2МлЃЌFeдЊЫиЕФЛЏКЯМлгЩ+2МлЩ§ИпЮЊ+3МлЁЂSдЊЫиЕФЛЏКЯМлгЩ-2МлЩ§ИпЮЊ+4МлЃЛ
(2)ЬњРызггіKSCNШмвКЮЊбЊКьЩЋЃЛ
(3)ЖўбѕЛЏСђЮЊЛЗОГЮлШОЮяЃЌВЛФмХХЗХЃЌПЩгУгкжЦБИСђЫсЛђбЧСђЫсбЮЃЛ
(4)ЖўбѕЛЏСђОпгаЦЏАзадЃЌЧвЦЏАзОпгаднЪБадЃЛ
(5)ЪЕбщЪвжЦБИЃЌЪеМЏИЩдяЕФSO2ЃЌгЩзАжУПЩжЊЃЌAЮЊЗДгІзАжУЃЌCЮЊИЩдязАжУЃЌBЮЊЪеМЏзАжУЃЌDЮЊЗРжЙЕЙЮќЃЌEЮЊЮВЦјДІРэЃЌвдДЫРДНтД№ЁЃ
(1)8CuFeS2+21O2 ![]() 8Cu+4FeO+2Fe2O3+16SO2жаЃЌCuдЊЫиЕФЛЏКЯМлгЩ+2МлНЕЕЭЮЊ0ЁЂOдЊЫиЕФЛЏКЯМлгЩ0НЕЕЭЮЊ-2МлЃЌЗДгІжаБЛЛЙдЕФдЊЫиЪЧCuЁЂOЃЛ
8Cu+4FeO+2Fe2O3+16SO2жаЃЌCuдЊЫиЕФЛЏКЯМлгЩ+2МлНЕЕЭЮЊ0ЁЂOдЊЫиЕФЛЏКЯМлгЩ0НЕЕЭЮЊ-2МлЃЌЗДгІжаБЛЛЙдЕФдЊЫиЪЧCuЁЂOЃЛ
(2)гУЯЁH2SO4НўХнЩЯЪіЗДгІжЦШЁЭКѓЪЃгрЕФЙЬЬхВадќЃЌШЁЩйСПЫљЕУШмвКЃЌМьбщШмвКжаДцдкFe3+ЕФЗНЗЈЪЧШЁЩйСПШмвКЃЌЕЮМгKSCNШмвКЃЌШмвКБфКьЃЛ
(3)ЖўбѕЛЏСђЮЊЛЗОГЮлШОЮяЃЌВЛФмИпПеХХЗХЃЌгыХЈСђЫсВЛЗДгІЃЌПЩгУгкжЦБИСђЫсЛђбЧСђЫсбЮЃЌжЛгаbcЗћКЯЃЛ
(4)бщжЄЛЦЭПѓвБСЖЭЕФЗДгІКѓЦјЬхжаКЌгаSO2ЕФЗНЗЈЪЧНЋЦјЬхЭЈШыЦЗКьШмвКжаЃЌШчЙћЦЗКьШмвКЭЪЩЋЃЌМгШШКѓгжБфКьЃЌдђжЄУїгаSO2ЃЛ
(5)ЂйзАжУAВњЩњSO2ЃЌЗДгІЕФЛЏбЇЗНГЬЪНЮЊCu+2H2SO4(ХЈ)![]() CuSO4+SO2Ёќ+2H2OЃЛ
CuSO4+SO2Ёќ+2H2OЃЛ
ЂкгЩзАжУПЩжЊЃЌAЮЊЗДгІзАжУЃЌCЮЊИЩдязАжУЃЌBЮЊЪеМЏзАжУЃЌDЮЊЗРжЙЕЙЮќЃЌEЮЊЮВЦјДІРэЃЌдђАДЦјСїЗНЯђСЌНгИївЧЦїНгПкЃЌЫГађЮЊaЁњdЁњeЁњcЁњfЃЌзАжУDЕФзїгУЪЧ АВШЋЦПЃЌЗРжЙЕЙЮќЃЛзАжУEжаNaOHШмвКЕФзїгУЪЧЮќЪеЖргрЕФSO2ЃЌЗРжЙЮлШОПеЦјЁЃ

ЁОЬтФПЁПдквЛЖЈЬхЛ§ЕФУмБеШнЦїжаЃЌНјааШчЯТЛЏбЇЗДгІЃКCO2(g)ЃЋH2(g) ![]() CO(g)ЃЋH2O(g)ЃЌЦфЛЏбЇЦНКтГЃЪ§KКЭЮТЖШtЕФЙиЯЕШчЯТБэЫљЪОЃК
CO(g)ЃЋH2O(g)ЃЌЦфЛЏбЇЦНКтГЃЪ§KКЭЮТЖШtЕФЙиЯЕШчЯТБэЫљЪОЃК
t Ёц | 700 | 800 | 830 | 1 000 | 1 200 |
K | 0.6 | 0.9 | 1.0 | 1.7 | 2.6 |
ЛиД№ЯТСаЮЪЬтЃК
ЃЈ1ЃЉИУЗДгІЕФЛЏбЇЦНКтГЃЪ§БэДяЪНЮЊKЃН_________________________________ЁЃ
ЃЈ2ЃЉИУЗДгІЮЊ________ЗДгІ(ЬюЁАЮќШШЁБЛђЁАЗХШШЁБ)ЁЃ
ЃЈ3ЃЉФГЮТЖШЯТЃЌИїЮяжЪЕФЦНКтХЈЖШЗћКЯЯТЪНЃК3c(CO2)ЁЄc(H2)ЃН5c(CO)ЁЄc(H2O)ЃЌЪдХаЖЯДЫЪБЕФЮТЖШЮЊ______ЁЃ
ЃЈ4ЃЉШє830 ЁцЪБЃЌЯђШнЦїжаГфШы1 mol COЁЂ5 mol H2OЃЌЗДгІДяЕНЦНКтКѓЃЌЦфЛЏбЇЦНКтГЃЪ§K______1.0(ЬюЁАДѓгкЁБЁАаЁгкЁБЛђЁАЕШгкЁБ)ЁЃ
ЃЈ5ЃЉ830 ЁцЪБЃЌШнЦїжаЕФЗДгІвбДяЕНЦНКтЁЃдкЦфЫћЬѕМўВЛБфЕФЧщПіЯТЃЌРЉДѓШнЦїЕФЬхЛ§ЁЃЦНКт____вЦЖЏ(ЬюЁАЯђе§ЗДгІЗНЯђЁБЁАЯђФцЗДгІЗНЯђЁБЛђЁАВЛЁБ)ЁЃ
ЃЈ6ЃЉШє1 200 ЁцЪБЃЌдкФГЪБПЬЦНКтЬхЯЕжаCO2ЁЂH2ЁЂCOЁЂH2OЕФХЈЖШЗжБ№ЮЊ2 molЁЄLЃ1ЁЂ2 molЁЄLЃ1ЁЂ4 molЁЄLЃ1ЁЂ4 molЁЄLЃ1ЃЌдђДЫЪБЩЯЪіЗДгІЕФЦНКтвЦЖЏЗНЯђЮЊ__________(ЬюЁАе§ЗДгІЗНЯђЁБЁАФцЗДгІЗНЯђЁБЛђЁАВЛвЦЖЏЁБ)ЁЃ