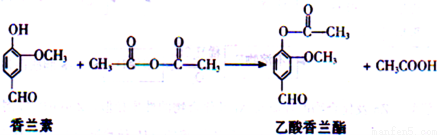
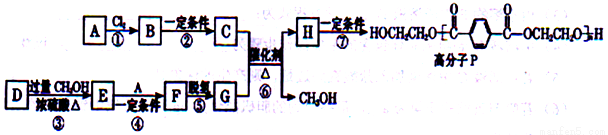
��Ŀ����
�����뻯ѧ�����йص�˵����ѧ���ﲻ��ȷ���ǣ� ��
A. ��ʯī���缫���NH4Cl��Һ��������ӦʽΪ��2Cl����2e����Cl2��
B. ��Al��Cu��Ũ������ɵ�ԭ��أ���������ӦʽΪ��NO3����2H����e��= NO2����H2O
C. ��Al��Mg��NaOH��ɵ�ԭ��أ��为����ӦʽΪ��Al��3e��+3OH�� = Al(OH)3��
D. Mg(OH)2��������Һ�д����ܽ�ƽ�⣺ Mg(OH)2(s)  Mg2+(aq) + 2OH��(aq)���ù��������NH4Cl��Һ
Mg2+(aq) + 2OH��(aq)���ù��������NH4Cl��Һ
��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ