��Ŀ����
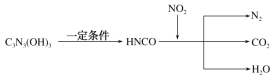
����Ŀ����(Sb)���仯�����ڹ�ҵ����������;���Ի����(��Ҫ�ɷ�ΪSb2S3��������PbS��As2S3��CuO��SiO2��)Ϊԭ���Ʊ�������Ĺ���������ͼ��ʾ��

��֪���� ����Һ�г������������SbCl5֮�⣬������SbCl3��PbCl2��AsCl3��CuCl2�ȣ�
�ڳ����£�Ksp(CuS)=1.27��10-36��Ksp(PbS)=9.04��10-29��
����Һ������Ũ��С�ڵ���1.0��10-5mol��L-1ʱ����Ϊ�����ӳ�����ȫ��
(1)����1�г���S֮�⣬����___________(�ѧʽ)��
(2)��������ʱ��Sb2S3������Ӧ�Ļ�ѧ����ʽΪ_________________��
(3)����ԭ��ʱ����Sb��ԭ������Ϊ_____________(�ѧʽ)��
(4)�����£�����ͭ��Ǧ��ʱ��Cu2+��Pb2+��������ȫ����ʱ��Һ�е�c(S2-)������______������Na2SҲ���˹��࣬��ԭ��Ϊ_____________��
(5)��������ʱ��H3PO3���ɣ��÷�Ӧ�����ӷ���ʽΪ________________��
(6)�������ʱ����������SbԪ���뱻��ԭ��SbԪ�ص�����֮��Ϊ_______��
���𰸡�SiO2 Sb2S3��3SbCl5��5SbCl3��3S SbCl5 9.04��10-24 mol��L��1 ����H2S����Ⱦ�����������Sb2S3 2As3++3PO23-+3H++3H2O=2As+3H3PO3 3��2
��������
�����(��Ҫ�ɷ�ΪSb2S3��������PbS��As2S3��CuO��SiO2��)���������ȡ�õ���ȡҺ������Һ�г������������SbCl5֮�⣬������SbCl3��PbCl2��AsCl3��CuCl2�ȣ�����1�г������ɵ�S֮���δ�ܽ�Ķ������裬����Һ�м���Sb��ԭSbCl5������SbCl3������Na2Sʱ��֤Cu2+��Pb2+��������ȫ�����˵õ�����2ΪCuS��PbS����Һ�м���Na3PO2 ��Һ���飬����H3PO3���鵥�ʣ�ʣ��SbCl3��Һ��ͨ����õ�Sb���ݴ˷������
(1)������������������1�г���S֮�⣬����SiO2���ʴ�Ϊ��SiO2��
(2)��������ʱ��Sb2S3����Һ�к�SbCl5����������ԭ��Ӧ�����ɵ���������Ȼ��࣬��Ӧ�Ļ�ѧ����ʽΪ��Sb2S3+3SbCl5=5SbCl3+3S���ʴ�Ϊ��Sb2S3+3SbCl5=5SbCl3+3S��
(3)��������ͼ��������������Sb��ԭ������Ϊ��̬SbCl5���ʴ�Ϊ��SbCl5��
(4)�����£�Ksp(CuS)=1.27��10-36��Ksp(PbS)=9.04��10-29�������£�����ͭ��Ǧ��ʱ��Cu2+��Pb2+��������ȫ���ܶȻ�������֪����Ǧȫ������ʱ��ͭ������ȫ����Һ������Ũ��С�ڵ���1.0��10-5molL-1ʱ����Ϊ�����ӳ�����ȫ����ʱ��Һ�е�c(S2-)��![]() =
=![]() =9.04��10-24 molL-1������Na2SҲ���˹��࣬��ԭ��Ϊ����H2S����Ⱦ�����������Sb2S3���ʴ�Ϊ��9.04��10-24molL-1������H2S����Ⱦ�����������Sb2S3��
=9.04��10-24 molL-1������Na2SҲ���˹��࣬��ԭ��Ϊ����H2S����Ⱦ�����������Sb2S3���ʴ�Ϊ��9.04��10-24molL-1������H2S����Ⱦ�����������Sb2S3��
(5)��Һ�м���Na3PO2 ��Һ���飬����H3PO3���鵥�ʣ���Ӧ�����ӷ���ʽΪ2As3++3PO23-+3H++3H2O=2As+3H3PO3���ʴ�Ϊ�� 2As3++3PO23-+3H++3H2O=2As+3H3PO3��
(6)����ͼ�е��ʱSbCl3��Ӧ����SbCl5��Sb�����ݵ����غ㣬Sb3+��Sb5+��2e-��Sb3+��Sb��3e-����������SbԪ���뱻��ԭ��SbԪ�����ʵ���֮��Ϊ3��2���ʴ�Ϊ��3��2��