��Ŀ����
��9�֣�1 Lij�����Һ�����ܺ��е� �������±���
�������±���
(1)������Һ����μ���NaOH��Һ���ʵ����ȣ�������������������ʵ����� �����NaOH��Һ�������
�����NaOH��Һ�������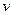 ���Ĺ�ϵ����ͼ��ʾ�������Һ��ȷ�����е�������_______________��
���Ĺ�ϵ����ͼ��ʾ�������Һ��ȷ�����е�������_______________��
����ȷ���Ƿ��е���������__________��Ҫȷ������ڿɲ�������ʵ����________��

��2������⣬����Һ�к��д�����Cl�� ��Br����I��������1 L�û����Һ��ͨ�롪������Cl2����Һ��Cl����Br����I�������ʵ�����ͨ��Cl2�������״������ ��ϵ���±���ʾ��������ش��������⣺
��ϵ���±���ʾ��������ش��������⣺
�ٵ�ͨ��Cl2�����Ϊ2.8 Lʱ����Һ�з�����Ӧ�����ӷ���ʽΪ___ ��
��ԭ��Һ��Cl����Br����I�������ʵ���Ũ��֮��Ϊ_____________��
 �������±���
�������±���| ���ܴ������е������� | H+ NH4+ Al3+ K+ |
| ���ܴ������е������� | Cl- Br- I? ClO? AlO2- |
 �����NaOH��Һ�������
�����NaOH��Һ�������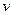 ���Ĺ�ϵ����ͼ��ʾ�������Һ��ȷ�����е�������_______________��
���Ĺ�ϵ����ͼ��ʾ�������Һ��ȷ�����е�������_______________������ȷ���Ƿ��е���������__________��Ҫȷ������ڿɲ�������ʵ����________��

��2������⣬����Һ�к��д�����Cl�� ��Br����I��������1 L�û����Һ��ͨ�롪������Cl2����Һ��Cl����Br����I�������ʵ�����ͨ��Cl2�������״������
 ��ϵ���±���ʾ��������ش��������⣺
��ϵ���±���ʾ��������ش��������⣺| Cl2���������״���� | 2.8L | 5.6 L | 11.2 L |
 ��Cl���� ��Cl���� | 1.25mol | 1.5 mol | 2 mol |
 ��Br���� ��Br���� | 1.5 mol | 1.4 mol | 0.9 mol |
 ��I���� ��I���� | 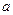 mol mol | 0 | 0 |
��ԭ��Һ��Cl����Br����I�������ʵ���Ũ��֮��Ϊ_____________��
��1��H+��Al3+��NH4+��3�֣� K����1�֣���ɫ��Ӧ��1�֣�
��2����Cl2��2I����I2��2Cl����2�֣� �� 10��15��4��2�֣�
��2����Cl2��2I����I2��2Cl����2�֣� �� 10��15��4��2�֣�
��

��ϰ��ϵ�д�
 ���ɶ���ܲ��¿�ֱͨ��Уϵ�д�
���ɶ���ܲ��¿�ֱͨ��Уϵ�д�
�����Ŀ
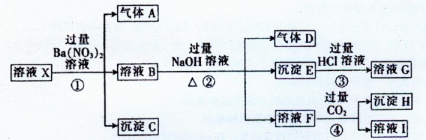
 M�Ļ��ϼ�Ϊ�� ��
M�Ļ��ϼ�Ϊ�� �� H++A2������֪0.1mol��L��1NaHA��Һ��pH = 2��������˵������ȷ����
H++A2������֪0.1mol��L��1NaHA��Һ��pH = 2��������˵������ȷ����