��Ŀ����
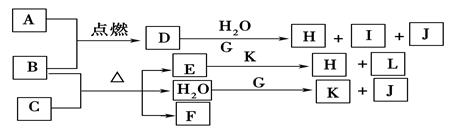
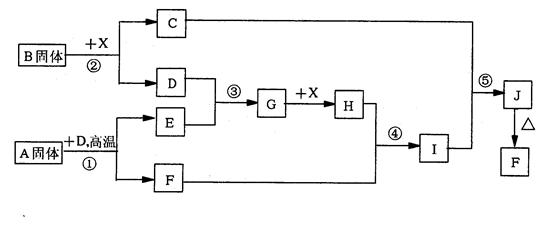
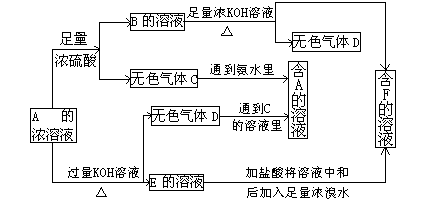
(10��)��ͼ��A~J�ֱ�����йط�Ӧ�е�һ�����ʣ����Ǿ�Ϊ��ѧ��ѧ�еij������ʡ���֪A~E��F~J�зֱ���ͬһ��Ԫ�ء���ӦE��A+O2������δ�����
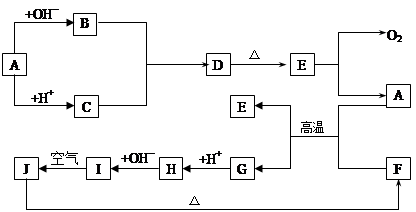
����д���пհף�
��1��������H�е��������� ��D�Ļ�ѧʽΪ ��
��2��A����B��Ӧ�����ӷ���ʽ�� ��
��3��A��F�Ļ�������Ӧ�Ļ�ѧ����ʽ�� ��
��4��д��B��C����D�����ӷ���ʽ ��
��5��I��J��Ӧ�Ļ�ѧ����ʽ�� ��
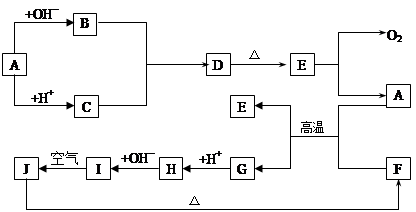
����д���пհף�
��1��������H�е��������� ��D�Ļ�ѧʽΪ ��
��2��A����B��Ӧ�����ӷ���ʽ�� ��
��3��A��F�Ļ�������Ӧ�Ļ�ѧ����ʽ�� ��
��4��д��B��C����D�����ӷ���ʽ ��
��5��I��J��Ӧ�Ļ�ѧ����ʽ�� ��
(10��)��1��Fe2+ Al��OH��3 ��2��2Al+2OH��+2H2O=2AlO2��+3H2��
��3�� 2Al+Fe2O3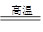 2Fe+Al2O3 ��4��Ai3++3AlO2��+6H2O=4Al��OH��3��
2Fe+Al2O3 ��4��Ai3++3AlO2��+6H2O=4Al��OH��3��
��5��4Fe��OH��2+O2+2H2O ===4Fe��OH��3
��3�� 2Al+Fe2O3
 2Fe+Al2O3 ��4��Ai3++3AlO2��+6H2O=4Al��OH��3��
2Fe+Al2O3 ��4��Ai3++3AlO2��+6H2O=4Al��OH��3����5��4Fe��OH��2+O2+2H2O ===4Fe��OH��3
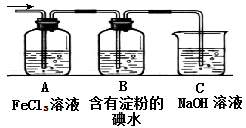
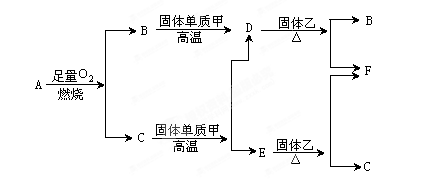
������������ݿ�ͼ��֪��A��ǿ�ᡢǿ���Ӧ�������ɵ�B��C�ַ�Ӧ����D����D�ֽ�����E��E�ֿ�������������A����˵��AӦ���ǵ�������B��ƫ�����ƣ�C�������Ȼ��������߷�Ӧ���ɰ�ɫ�����������������������ֽ�������������������������������͵�����������A��F��Ӧ����E��G�ķ�Ӧ�������жϣ��÷�Ӧ���������ȷ�Ӧ����G�Ϳ��������������ᷴӦ�����������ӣ����������ں�ǿ�Ӧ�����������������������������ױ��������������������������������ֽ�������������������F����������J������������I��������������
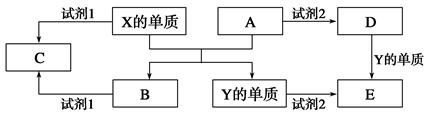
��������ѧ�ƶ�����һ���ۺ��Խ�ǿ�����⣬��Ԫ�ؼ����������ʺ�������������������ѧ�����֪ʶ����������ѧ�Ƽ��ۺϡ��������ɿ���ѧ���Ի�ѧ֪ʶ������̶ȣ�����Ҫ��������ѧ�����ۺϷ���������˼ά���������ͼ��ķ�������ؼ�����Ѱ�ҡ�ͻ�ƿڡ�����ͻ�ƿڡ�����ץ���ء��֣�����������ɫ������״̬��������ζ�����ⷴӦ���������������Ʒ���������;�ȡ�

��ϰ��ϵ�д�
 �߽�������ϵ�д�
�߽�������ϵ�д�
�����Ŀ
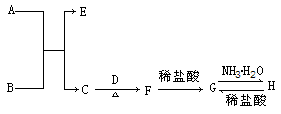

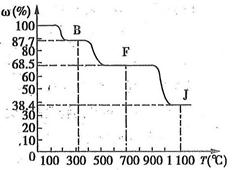
 (��)]���¶�[T(��)]�ı仯���ߡ�
(��)]���¶�[T(��)]�ı仯���ߡ�