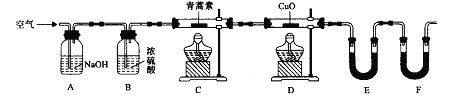
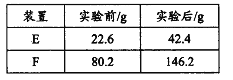
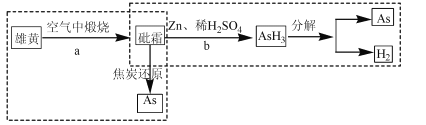
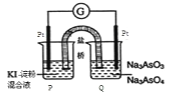
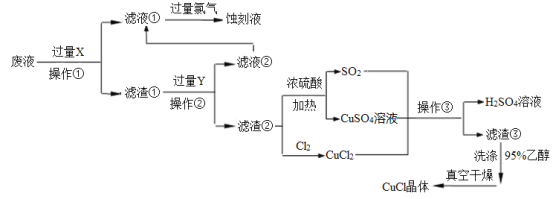
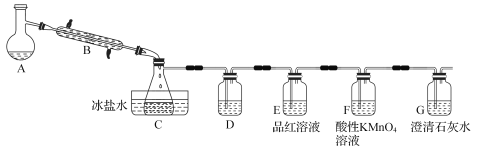
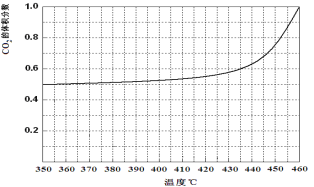
��Ŀ����
����Ŀ��50 mL 0.50 mol��L-1��������50 mL 0.55 mol��L-1NaOH��Һ����ͼ��ʾ��װ���н����кͷ�Ӧ��ͨ���ⶨ��Ӧ���������ų��������ɼ����к��ȣ�����������⣺

��1����ʵ��װ���Ͽ���ͼ����ȱ�ٵ�һ�ֲ�����Ʒ��_____________����ָ��װ���е���������� ��
��2���ձ���������ֽ����������___________��
��3�����ձ����粻��Ӳֽ�壬��õ��к�����ֵ___________������ƫ������ƫС������Ӱ��������
��4��ʵ���и���60mL0.50mol��L-1�������60mL0.55mol��L-1NaOH��Һ��������ʵ����ȣ����ų�������___________(���������������������������к���___________������������������������
��5������ͬŨ������İ�ˮ����NaOH��Һ��������ʵ�飬��õ��к��ȵ���ֵ��_____����50 mL 0.50 mol��L-1NaOH��Һ��������ʵ�飬��õ��к��ȵ���ֵ��_____��������ƫ������ƫС������Ӱ��������
��������1�����β������������С�ձ���û����ƽ��2����С��������е�������ʧ��3��ƫС��4������� ��ȣ�5��ƫС ƫС
��������
�����������1���ⶨ�к��ȣ�ʹ��Һ��Ͼ��ȣ�����ȱ�����β��������������װ��ͼ��֪װ���е���������Ǵ�С�ձ���û����ƽ��
��2��Ϊ����ʵ������е�������ʧ���ձ���������ֽ���Լ�С��������е�������ʧ��
��3�����ձ����粻��Ӳֽ������Ӧ�ų���������ʧ����õ��к�����ֵƫС��
��4�������60mL0.50mol/L������60mL 0.55mol/L NaOH��Һ���з�Ӧ�����ɵ�ˮ�����ʵ�������50mL 0.50mol/L������50mL 0.55mol/L NaOH�࣬������������к�����ָ����1molˮ�ų��������������������أ������к�����ȣ�
��5�����ᡢ����������ȣ�����ͬŨ�Ⱥ�����İ�ˮ��NH3H2O������NaOH��Һ��������ʵ�飬��õ��к��ȵ���ֵ��ƫС����50 mL 0.50 mol��L-1NaOH��Һ��������ʵ�飬���ܻ������������ȫ��Ӧ�������õ��к��ȵ���ֵ��ƫС��