��Ŀ����
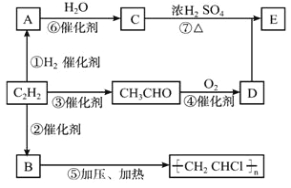
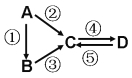
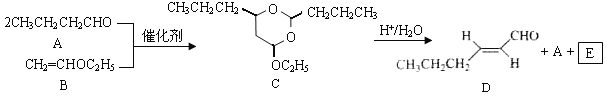
����Ŀ����-2-��ϩȩ��D����һ����Ҫ�ĺϳ����ϣ����кϳ�·�����Ʊ�D�ķ���֮һ�����ݸúϳ�·�ش��������⣺
��֪��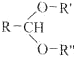
![]() RCHO+R��OH+R��OH
RCHO+R��OH+R��OH
��1��A��������__________��B�����й���ԭ����Ŀ���Ϊ__________��C�������뻷���������������У���ͬ��ѧ��������ԭ�ӹ���__________�֡�
��2��D�к��������ŵ�������__________��д������ù����ŵĻ�ѧ��Ӧ����ʽ__________��
��3��EΪ�л���ܷ����ķ�Ӧ��__________
a���ۺϷ�Ӧ b���ӳɷ�Ӧ c����ȥ��Ӧ d��ȡ����Ӧ
��4��B��ͬ���칹��F��B����ȫ��ͬ�Ĺ����ţ�д��F���п��ܵĽṹ________��
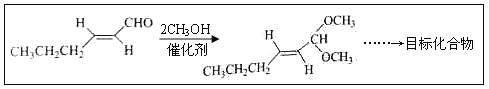
��5����DΪ��Ҫԭ���Ʊ���ȩ��Ŀ�껯���������ڷ����н��ϳ�·�ߵĺ�벿�ֲ���������
��6��������5���ĺϳ�·���е�һ����Ӧ��Ŀ����__________��
���𰸡�
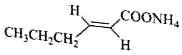
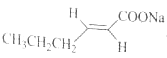
��1������ȩ��ȩ 9 8
��2��ȩ��
![]() +2Ag��NH3��2OH
+2Ag��NH3��2OH![]()
 +2Ag��+3NH3+H2O��
+2Ag��+3NH3+H2O��![]() +2Cu��OH��2+NaOH
+2Cu��OH��2+NaOH![]()
 +Cu2O��+3H2O��
+Cu2O��+3H2O��
��3��cd��
��4��CH2=CHCH2OCH3��![]() ��
��![]() ��
��![]() ��
��
��5��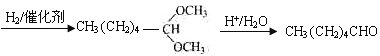 ��
��
��6������ȩ������������������
��������
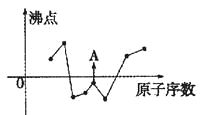
�����������1����������ͼ��AΪCH3CH2CH2CHO���Ƕ�ȩ��BΪCH2=CHOC2H5�������й���ԭ����Ŀ���Ϊ9������ͼ��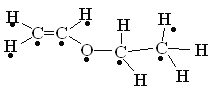 ����C�������뻷���������������У�8��̼ԭ���ϵ���ԭ�ӻ�ѧ����������ͬ������8�֣��ʴ�Ϊ����ȩ��9��8��
����C�������뻷���������������У�8��̼ԭ���ϵ���ԭ�ӻ�ѧ����������ͬ������8�֣��ʴ�Ϊ����ȩ��9��8��
��2��DΪ![]() �����к�����������ȩ��������ȩ��������������Һ������������ͭ����Һ���ʴ�Ϊ��ȩ����
�����к�����������ȩ��������ȩ��������������Һ������������ͭ����Һ���ʴ�Ϊ��ȩ����![]() +2Ag��NH3��2OH
+2Ag��NH3��2OH![]()
 +2Ag��+3NH3+H2O��
+2Ag��+3NH3+H2O��![]() +2Cu��OH��2+NaOH
+2Cu��OH��2+NaOH![]()
 +Cu2O��+3H2O��
+Cu2O��+3H2O��
��3����������ͼ�������Ϣ��C�����������·�Ӧ����![]() ��CH3CH2CH2CHO��CH3CH2OH�Լ�ˮ�����EΪCH3CH2OH�����ڴ����ܷ����ķ�Ӧ����ȥ��Ӧ��ȡ����Ӧ����ѡcd��
��CH3CH2CH2CHO��CH3CH2OH�Լ�ˮ�����EΪCH3CH2OH�����ڴ����ܷ����ķ�Ӧ����ȥ��Ӧ��ȡ����Ӧ����ѡcd��
��4��B��ͬ���칹��F��B����ȫ��ͬ�Ĺ����ţ� F���ܵĽṹ�У�CH2=CHCH2OCH3 ![]() ��
��![]() ��
��![]() ���ʴ�Ϊ��CH2=CHCH2OCH3
���ʴ�Ϊ��CH2=CHCH2OCH3 ![]() ��
��![]() ��
��![]() ��
��
��5��DΪ![]() ����ȩ�Ľṹ��ʽΪCH3CH2CH2CH2CH2CHO��������Ϣ�ͼ�ȩ�Ľṹ��
����ȩ�Ľṹ��ʽΪCH3CH2CH2CH2CH2CHO��������Ϣ�ͼ�ȩ�Ľṹ��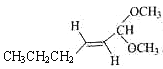 ������Ҫ��̼̼˫��ת��Ϊ������Ȼ�������������·�Ӧ���ɣ��ʴ�Ϊ��
������Ҫ��̼̼˫��ת��Ϊ������Ȼ�������������·�Ӧ���ɣ��ʴ�Ϊ�� ��
��
��6��ȩ��Ҳ�ܹ��������ӳɣ���5���кϳ�·���е�һ����Ӧ��Ŀ���DZ���ȩ�����ʴ�Ϊ������ȩ����

����Ŀ����ҵ��ˮ�г����в�ͬ���͵���Ⱦ�����ɲ��ò�ͬ�ķ������������´�����ʩ�ͷ�����ȷ����
ѡ�� | ��Ⱦ�� | ������ʩ | ������� |
A | ���� | ����ʯ���к� | ������ |
B | Cu2+���ؽ������� | �������γ��� | ��ѧ�� |
C | ������ˮ | �û���̿���� | ������ |
D | ������ķ�ˮ | ��ʯ��ˮ��Ӧ | ��ѧ�� |