题目内容
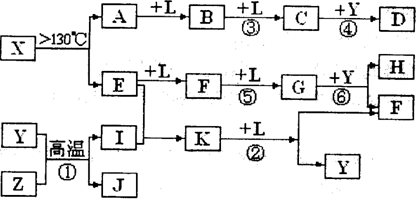
(10分)A、B、C、D、E、F六种物质的转化关系如图所示(反应条件和部分产物未标出)。

(1)若A为短周期金属单质,D为短周期非金属单质,且所含元素的原子序数A是D的2倍,所含元素的原子最外层电子数D是A的2倍,F的浓溶液与A、D反应都有红棕色气体生成,则C的电子式为 ,反应④的化学方程式为_______________________________________。
(2)若A是常见的变价金属的单质,D、F是气态单质,且反应①在水溶液中进行。请写出在水溶液中进行的反应②的离子方程式 ;
已知光照条件下D与F反应生成B,写出该反应的化学方程式: 。
(3)若A、D、F都是短周期非金属元素单质,且A、D所含元素同主族,A、F所含元素同周期,则反应①的化学方程式为 。

(1)若A为短周期金属单质,D为短周期非金属单质,且所含元素的原子序数A是D的2倍,所含元素的原子最外层电子数D是A的2倍,F的浓溶液与A、D反应都有红棕色气体生成,则C的电子式为 ,反应④的化学方程式为_______________________________________。
(2)若A是常见的变价金属的单质,D、F是气态单质,且反应①在水溶液中进行。请写出在水溶液中进行的反应②的离子方程式 ;
已知光照条件下D与F反应生成B,写出该反应的化学方程式: 。
(3)若A、D、F都是短周期非金属元素单质,且A、D所含元素同主族,A、F所含元素同周期,则反应①的化学方程式为 。
(1) 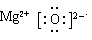
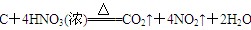
(2) 2Fe2++Cl2 === 2Fe3++2Cl-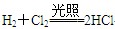
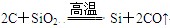
(3)
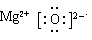
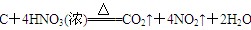
(2) 2Fe2++Cl2 === 2Fe3++2Cl-
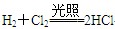
(3)

(1)红棕色气体是NO2,所以F是浓硝酸。①是置换反应,在常见金属置换非金属的反应中,除了活泼金属置换氢气外,就是镁在CO2中燃烧生成单质碳,方程式为2MgO+CO2 2MgO+C,即A是镁,B是CO2,C是MgO,D是碳。
2MgO+C,即A是镁,B是CO2,C是MgO,D是碳。
(2)常见的变价金属的单质是铁,根据框图可知A、C、E中均含有铁元素,且价态不同,所以F应是具有氧化性的氯气。即A、B、C、D、E、F分别为Fe、HCl、FeCl2、H2、FeCl3、Cl2。
(3)根据框图可知A、C、E中均含有A元素,且价态不同,所以可能是碳元素。又因为在高温下碳可以置换SiO2中的硅,方程式为SiO2+2C Si+2CO↑。即A、B、C、D、E、F分别为C、SiO2、CO、Si、CO2、O2。
Si+2CO↑。即A、B、C、D、E、F分别为C、SiO2、CO、Si、CO2、O2。
 2MgO+C,即A是镁,B是CO2,C是MgO,D是碳。
2MgO+C,即A是镁,B是CO2,C是MgO,D是碳。(2)常见的变价金属的单质是铁,根据框图可知A、C、E中均含有铁元素,且价态不同,所以F应是具有氧化性的氯气。即A、B、C、D、E、F分别为Fe、HCl、FeCl2、H2、FeCl3、Cl2。
(3)根据框图可知A、C、E中均含有A元素,且价态不同,所以可能是碳元素。又因为在高温下碳可以置换SiO2中的硅,方程式为SiO2+2C
 Si+2CO↑。即A、B、C、D、E、F分别为C、SiO2、CO、Si、CO2、O2。
Si+2CO↑。即A、B、C、D、E、F分别为C、SiO2、CO、Si、CO2、O2。
练习册系列答案
相关题目
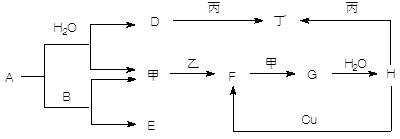



 。
。 。
。