��Ŀ����
5���±���Ԫ�����ڱ�һ���֣���Ա��Т١�����Ԫ�أ���ش��й����⣺���� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
| 2 | �� | �� | �� | |||||
| 3 | �� | �� | �� | �� | �� | |||
| 4 | �� | �� |
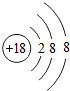 ��
����2���������γ��������������Ԫ����Al����Ԫ�ط��ű�ʾ����д����Ԫ�صĵ����������������ˮ���ﷴӦ�Ļ�ѧ����ʽ2Al+2NaOH+2H2O=2NaAlO2+3H2����
��3 ��������������ˮ�������ʽ��
 �û������������ӣ�����ۡ������ӡ��������
�û������������ӣ�����ۡ������ӡ����������4����������������ˮ�����У�������ǿ����HClO4���ѧʽ��
��5����Ԫ�����Ԫ�����ߺ˵����֮����26��
��6���ڢ����ĵ����У���ѧ���ʽϻ��õ���������������������ʲô��ѧ��Ӧ˵������ʵ��д����Ӧ�����ӷ���ʽ����Cl2+2Br-=2Cl-+Br2��
���� ��Ԫ�������ڱ���λ�ã���֪��ΪC����ΪN����ΪF����ΪNa����ΪAl����ΪS����ΪCl����ΪAr����ΪK����ΪBr��
��1�����л�ѧ��������õ�Ԫ����ϡ����Ԫ��Ar��
��2���������γ��������������Ԫ����Al�������������Ʒ�Ӧ����ƫ�����ƺ�������
��3 ��������������ˮ����Ϊ�������أ��û��������ɼ����Ӻ�������������ɣ�
��4��ͬ����������ҽ����Լ������ǽ�������ǿ��ͬ�������϶��½�������ǿ���ǽ����Լ������ǽ���ǿ��Ԫ�أ�����������ˮ���������Ҳǿ��
��5��FΪ9��Ԫ�أ�BrΪ35��Ԫ�أ��ݴ˴��⣻
��6���ȵķǽ�����ǿ���壬���������Ļ�����ǿ���壬�����������û����廯�����Һ�е��壮
��� �⣺��Ԫ�������ڱ���λ�ã���֪��ΪC����ΪN����ΪF����ΪNa����ΪAl����ΪS����ΪCl����ΪAr����ΪK����ΪBr����1�����л�ѧ��������õ�Ԫ����ϡ����Ԫ��Ar����ԭ�ӽṹʾ��ͼΪ ��
��
�ʴ�Ϊ�� ��
��
��2���������γ��������������Ԫ����Al�������������Ʒ�Ӧ����ƫ�����ƺ���������Ӧ�Ļ�ѧ����ʽΪ2Al+2NaOH+2H2O=2NaAlO2+3H2����
�ʴ�Ϊ��Al�� 2Al+2NaOH+2H2O=2NaAlO2+3H2����
��3 ��������������ˮ����Ϊ�������أ������ʽΪ���û��������ɼ����Ӻ�������������ɣ����������ӻ����
�ʴ�Ϊ�� �� ���ӣ�
�� ���ӣ�
��4��ͬ����������ҽ����Լ������ǽ�������ǿ��ͬ�������϶��½�������ǿ���ǽ����Լ������ǽ���ǿ��Ԫ�أ�����������ˮ���������Ҳǿ����û������������ˮ������Ա�������������ˮ�����У�������ǿ����HClO4��
�ʴ�Ϊ��HClO4��
��5��FΪ9��Ԫ�أ�BrΪ35��Ԫ�أ����ߺ˵����֮����26��
�ʴ�Ϊ��26��
��6���ȵķǽ�����ǿ���壬���������Ļ�����ǿ���壬�����������û����廯�����Һ�е��壬��Ӧ�����ӷ���ʽΪCl2+2Br-=2Cl-+Br2��
�ʴ�Ϊ��������Cl2+2Br-=2Cl-+Br2��
���� ���⿼��Ԫ�����ڱ���Ԫ���������ۺ�Ӧ�ã��Ƚϻ����������ڻ���֪ʶ�Ĺ��̣�

| ���� | CH3COOH | H2CO3 | HNO2 |
| ���볣����25�棩 | K=1.8��10-5 | K1=4.3��10-7 K2=5.6��10-11 | K=5.0��10-4 |
��1�����¶��£���0.1mol��L-1CH3COOH ��Һ�м�������CH3COONa ����ʱ������˵����ȷ����ad������ţ���
a����Һ��pH���� b�� CH3COOH�ĵ���̶�����
c����Һ�ĵ����������� d����Һ��c��OH- ��•c��H+ ������
��2��25��ʱ��0.10mol��L-1 Na2CO3 ��Һ��pH=11������Һ����ˮ�������c��OH- ����10-3mol��L-1
��3�������£���20mL 0.10mol��L-1 CH3COOH��Һ��20mL 0.10mol��L-1 HNO2 ��Һ�ֱ���20mL 0.10mol��L-1 NaHCO3 ��Һ��ϣ���Ϻ���Һ����仯���Բ��ƣ���
�ٷ�Ӧ��ʼʱ��v��CH3COOH ���� v��HNO2�������������������������ԭ���Ǿ������ƽ�ⳣ����֪��HNO2�����Աȴ���ǿ����������Ա�̼��ǿ����ͬ���ʵ���Ũ�ȵĴ����HNO2��ȣ�HNO2��Һ��������Ũ�ȴ�������̼��������Һ��Ӧ���ʿ죮
�ڳ�ַ�Ӧ������Һ��c��CH3COO-����c��NO2-�������������������=������
��4��25��ʱ����CH3COOH��Һ�м���һ������NaHCO3�����û��Һ��pH=6������Һ��$\frac{c��C{H}_{3}CO{O}^{-}��}{c��C{H}_{3}COOH��}$=18��
| A�� | 30 g•mol-1��46 g•mol-1 | B�� | 44 g•mol-1��80 g•mol-1 | ||
| C�� | 28 g•mol-1 | D�� | 46 g•mol-1 |
| A�� | ��KIO3����������Һ�е�KI��5I-+IO${\;}_{3}^{+}$+3H2O�T3I2+6OH- | |
| B�� | ��NH4HSO3��Һ�мӹ�����NaOH��Һ�����ȣ�NH${\;}_{4}^{+}$+OH-$\frac{\underline{\;\;��\;\;}}{\;}$NH3��+H2O | |
| C�� | ������SO2ͨ���䰱ˮ�У�SO2+NH3•H2O�THSO${\;}_{3}^{-}$+NH${\;}_{4}^{+}$ | |
| D�� | Ba��OH��2��Һ����μ���һ�����ʵ���Ũ�ȵ�NaHSO4��Һ������Һ�е�OH-ǡ�÷�Ӧһ��ʱ��Ba2++2OH-+2H++SO${\;}_{4}^{2-}$�TBaSO4��+2H2O |
| A�� | 2C2H2��g��+5O2��g���T4CO2��g��+2H2O��l����H�T-2599.2 kJ•mol-1 | |
| B�� | 2C2H2��g��+5O2��g���T4CO2��g��+2H2O��l����H�T-389.9 kJ•mol-1 | |
| C�� | C2H2��g��+5/2 O2��g���T2CO2��g��+H2O��l����H�T-1299.6 kJ•mol-1 | |
| D�� | 2C2H2��g��+5O2��g���T4CO2��g��+2H2O��g����H�T-1299.6kJ•mol-1 |
| A�� | ��ƽ������ӷ���ʽΪ2O3+2I-+4H+=I2+2O2+2H2O | |
| B�� | ÿ����1 mol I2ת�Ƶ���2 mol | |
| C�� | O2�ǻ�ԭ����֮һ | |
| D�� | �÷�Ӧ��˵��O2�������Դ���I2�� |
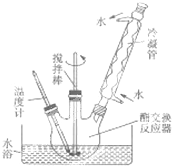 ��������Ǹ�֬���������������֬��״�ͨ��ȡ����Ӧ���������������������´����õ����ò������Ʊ�������͵IJ������£�
��������Ǹ�֬���������������֬��״�ͨ��ȡ����Ӧ���������������������´����õ����ò������Ʊ�������͵IJ������£�