��Ŀ����
5��2010���Ϻ������������ǡ����У�����������á����Իش��������⣺��1��Ϊȷ���������ڼ���ǿ������������ʴﵽ95%���ϣ��������ڼ�Ŀ�������״������У�����Ҫ����ָ����d��
a������������PM10��b��NO2Ũ�� c��SO2Ũ�� d��CO2Ũ�ȣ�
��2��Ϊ�˸��ƿ���������������ƴ����ж�������������̳�����Ⱦ����ŷ�������������װβ��������װ�ã�ʹ���е��к�����NO��COת��Ϊ�����壬�÷�Ӧ�Ļ�ѧ����ʽΪ2NO+2CO$\frac{\underline{\;����\;}}{\;}$N2+2CO2��
��3��Ϊ2010���������ڼ��ṩ��ѧ��Ч�Ļ����������Ϸ�������������ˮ�������������죬������ˮʱ����KAl��SO4��2•12H2O���ʽ�Ȼ����������ǻ�������ͨ�������������ȵ���������������ijũ�����Ϊ�������ˮ���ڽ��ر�ˮȡ�ؼҺ�ʹ��Ư�ۻ�Ư��Ƭ����ɱ����������ԭ�����û�ѧ����ʽ��ʾΪCa��ClO��2+H2O+CO2=CaCO3��+2HClO��
��4�����գ��������й���--������֮�ڡ������ɸֽ��������7000����ɫ�����1200��鲣���Ƚ��ɣ����в��ֲ���ΪʯӢ����������ѧ�ȶ���ǿ������ϵ��С����һ�����ֲ�����ʯӢ��������Ҫ�ɷ���SiO2��
���� ��1�����������ձ��м����ǿ����������Ͷ�������͵������
��2��һ��������һ����̼�ڴ��������·�Ӧ���ɵ����Ͷ�����̼��
��3����������������������������������������Ͷ���������ǿ������������������Ư���ܺͶ�����̼��ˮ��Ӧ���ɴ����ᣬ��������������
��4��ʯӢ��������Ҫ�ɷ���SiO2��
��� �⣺��1�����������ձ��м����ǿ����������Ͷ�������͵���������Զ�����̼���ڼ�ⷶΧ֮�ڣ���ѡd��
��2��һ��������һ����̼�ڴ��������·�Ӧ���ɵ����Ͷ�����̼����Ӧ����ʽΪ2NO+2CO$\frac{\underline{\;����\;}}{\;}$N2+2CO2���ʴ�Ϊ��2NO+2CO$\frac{\underline{\;����\;}}{\;}$N2+2CO2��
��3����������ǿ����������ˮ�����������������壬��������������������ԣ�������ˮ�е����������������ˮ����K2SO4•Al2��SO4��3•24H2O���ʽ�Ȼ����������ǻ������������Ͷ���������ǿ��������ɱ����������������������������ƺͶ�����̼��ˮ��Ӧ����̼��ƺʹ����ᣬ��Ӧ����ʽΪCa��ClO��2+H2O+CO2=CaCO3��+2HClO��
�ʴ�Ϊ������������������Ca��ClO��2+H2O+CO2=CaCO3��+2HClO��
��4��ʯӢ��������Ҫ�ɷ���SiO2���ʴ�Ϊ��SiO2��
���� ���⿼���˳������������Ⱦ���������ѶȲ���ע�������̼���ڿ��������ձ��ļ�ⷶΧ�У�ע�����֪ʶ�Ļ��ۣ�

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�| A�� | ����£�ȼ��1mol S���յ�����Ϊ297.23 kJ | |
| B�� | S ��g��+O2 ��g���TSO2 ��g�� �ų�����������297.23 kJ | |
| C�� | S ��g��+O2 ��g���TSO2 ��g���ų�������С��297.23 kJ | |
| D�� | �γ�1 mol SO2 �Ļ�ѧ�����ͷŵ����������ڶ��� 1 molS ��s���� 1mol O2 ��g���Ļ�ѧ�������յ������� |
��2����Ӫ�����⼰ѧ�����巢����Ҫ�ĽǶȿ��ǣ�����Ϊ��ͼʳ����Ӧ���Ӹ��������ʵ�ʳ�����㣨����һ�֣���
| ��ʳ | �� |
| ��ʳ | �������ײˡ��ܲ��� |
| A�� | ��Al��Mg��ϡ������Һ��ɵ�ԭ��أ��为����ӦʽΪ��Al-3e-�TAl3+ | |
| B�� | ��Al��Mg��NaOH��Һ��ɵ�ԭ��أ��为����ӦʽΪ��Al-3e-+3OH-�TAl��OH��3�� | |
| C�� | ��Fe��Zn��NaCl��Һ��ɵ�ԭ��أ��为����ӦʽΪ��Fe-2e-�TFe2+ | |
| D�� | ��Al��Cu��Ũ������Һ��ɵ�ԭ��أ��为����ӦʽΪ��Cu-2e-�TCu2+ |
| A�� | һ��������3������ | B�� | �������һ���µ�ͬλ�� | ||
| C�� | ����ɿ���H3+��ʾ | D�� | ����һ����ͨ����Ӷ�һ����ԭ�� |
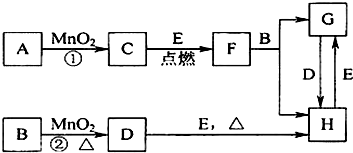
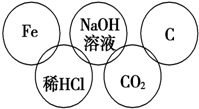 С������������������5�����ʣ����������ʼ��ܷ�����Ӧ�������������ʼ䲻�ܷ�����Ӧ��������������⣺
С������������������5�����ʣ����������ʼ��ܷ�����Ӧ�������������ʼ䲻�ܷ�����Ӧ��������������⣺