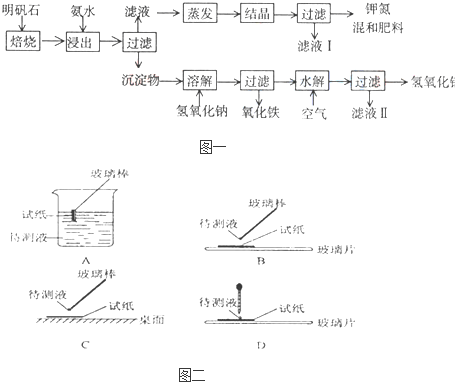
��Ŀ����
ij��ɫ����Һ���ܺ����������ӣ�K+��Al3+��Fe3+��Ba2+��������ɫʯ����ֽ������Һ����ֽ�Ժ�ɫ��
��ȡ��Һ����������ͭƬ��ϡ���Ṳ�ȣ�������ɫ���壬��������������������Ϊ����ɫ��
��ȡ��Һ���������백ˮ�а�ɫ�������ɣ��������������ˮ����������ʧ��
��ȡ��Һ�����������Ȼ�����Һ������ɫ������
��ȡʵ��ܺ�ij�����Һ��������������Һ������ɫ�������ټ��������ϡ���ᣬ��������ʧ��
��ش��������⣺
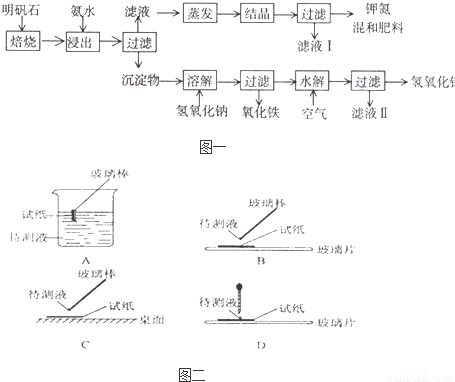
(1)��ʵ����У���ͼ��ʾ�IJ�������ȷ����__________(�����)��

(2)��������ʵ���ж�ԭ��Һ�п϶����ڵ�������__________���϶������ڵ�������__________��
(3)д����ڢ�����ʵ���йص����ӷ���ʽ��
��__________________________________________________________________
��___________________________________________________________________
(1)BD
(2)![]() ��Al3+��
��Al3+��![]() Fe3+��Ba2+��
Fe3+��Ba2+��![]()
(3)��3Cu+8H++2![]()
![]() 3Cu2++2NO��+4H2O
3Cu2++2NO��+4H2O
��Al3++3NH3��H2O![]() Al(OH)3��+3
Al(OH)3��+3![]()
�������ɡ���ɫ����Һ���ų�Fe3+�Ĵ��ڣ��ɢ�֪����Һ�����ԣ���![]() �����ڣ��ɢ�֪��һ������
�����ڣ��ɢ�֪��һ������![]() ���ɢ�֪��һ������Al3+���ɢ�֪��һ������
���ɢ�֪��һ������Al3+���ɢ�֪��һ������![]() ����Ba2+�����ڣ����ڢܼ������BaCl2�������ж�Cl-�Ƿ����.
����Ba2+�����ڣ����ڢܼ������BaCl2�������ж�Cl-�Ƿ����.

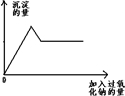 ij��ɫ����Һ�п��ܺ�Mg2+��Al3+��Fe3+��Cu2+��NH4+��K+�еļ������ӣ�����������Ĺ�������ʱ������ɫ��ζ�����������ͬʱ���ɰ�ɫ����������Ĺ������Ƶ����������������֮��Ĺ�ϵ����ͼ��ʾ���Իش�
ij��ɫ����Һ�п��ܺ�Mg2+��Al3+��Fe3+��Cu2+��NH4+��K+�еļ������ӣ�����������Ĺ�������ʱ������ɫ��ζ�����������ͬʱ���ɰ�ɫ����������Ĺ������Ƶ����������������֮��Ĺ�ϵ����ͼ��ʾ���Իش�