��Ŀ����
����Ŀ����0.1 mol/L��NaOH��Һ�ֱ�ζ������Ϊ20mL��Ũ�Ⱦ�Ϊ0.1 mol/L��HCl��Һ��HX��Һ����Һ��pH����� NaOH��Һ����ı仯��ͼ��ʾ������˵����ȷ����
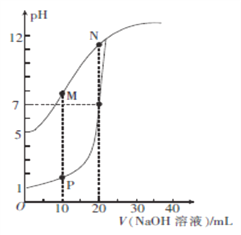
A. HX�ĵ���ƽ�ⳣ��KaԼΪ1��10-5
B. M���Ӧ��Һ��:c(HX)< c (X-)
C. P���Ӧ��Һ��: c(Cl-)=0.05mol/L
D. N���Ӧ��Һ��:c(Na+)��c(X-)��c(OH-) ��c(H+)
���𰸡�D
��������A.��ͼ��֪����HCl��ǿ�ᣬ0.1mol/L��HCl��ҺpH=1����0.1mol/L��HX��ҺpH=5��HX�����ᣬHX�ĵ���ƽ�ⳣ��Ka=![]() =1��10-9����A����B.M��ʱ����NaOH��Һ�����Ϊ10mL��������ҺΪ��Ũ�ȵ�HX��NaX�Ļ����Һ����ʱpH��7����Һ�ʼ�����˵��X����ˮ��̶ȴ���HX�ĵ���̶ȣ���c(HX)��c (X��)����B����C.p��ʱ����NaOH��Һ�����Ϊ10mL��������Һ��c(Cl��)=
=1��10-9����A����B.M��ʱ����NaOH��Һ�����Ϊ10mL��������ҺΪ��Ũ�ȵ�HX��NaX�Ļ����Һ����ʱpH��7����Һ�ʼ�����˵��X����ˮ��̶ȴ���HX�ĵ���̶ȣ���c(HX)��c (X��)����B����C.p��ʱ����NaOH��Һ�����Ϊ10mL��������Һ��c(Cl��)= ![]() =0.067mol/L����C����D.N��ʱ����NaOH��Һ�����Ϊ20mL��HX��NaOHǡ����ȫ��Ӧ��NaX��Һ��X������ˮ�ⷴӦʹ��Һ�ʼ��ԣ���c(Na+)��c(X��)��c(OH��) ��c(H+)����D��ȷ����ѡD��
=0.067mol/L����C����D.N��ʱ����NaOH��Һ�����Ϊ20mL��HX��NaOHǡ����ȫ��Ӧ��NaX��Һ��X������ˮ�ⷴӦʹ��Һ�ʼ��ԣ���c(Na+)��c(X��)��c(OH��) ��c(H+)����D��ȷ����ѡD��

����Ŀ��Ϊ��̽����������ˮ�Ļ�ԭ�ԣ�ij��ȤС��ͬѧ���������̽�����
I.̽�������Ļ�ԭ��
����ȤС��ͬѧ��������װ��(�г֣�����������)̽�������백���ķ�Ӧ������A��F�ֱ�Ϊ�����Ͱ����ķ���װ����BΪ��������������백����Ӧ��װ�á�
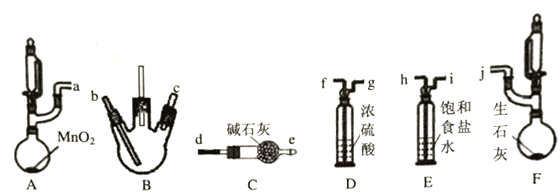
��ش�����������
��1������װ�ýӿڵ�����˳��Ϊa��h��i��f��g��___��____��___��____��j������װ��D��������____________��
(2)������������װ��B�г��ֵ�����Ϊ____________��
II.̽����ˮ�Ļ�ԭ��
����ȤС��ͬѧ̽����ͬ�����¸��������Һ�백ˮ�ķ�Ӧ��ʵ������:
ʵ�� | ���� | ���� |
�� | ȡ2mL.0.01mol/LKMnO4��Һ���Թ��У������¿���1mLŨ��ˮ�������ι�����ˮ��������Ƥ����ס�� | �����غ�ɫ����(MnO2),Լ10min����Һ�Ϻ�ɫ��dz |
�� | ȡ2mL0.01mol/LKMnO4��Һ���Թ��У������¿���1mLŨ��ˮ�������ι�1:5�����ᣬ������Ƥ����ס�� | �����غ�ɫ����(MnO2),��Һ�Ϻ�ɫ���̱�dz��Լ2min����Һ�Ϻ�ɫ��ȫ��ȥ |
�� | ȡ2mL0.1mol/LKMnO4��Һ���Թ��У������¿���ImLŨ��ˮ�������ι�����ˮ��������Ƥ����ס�� | �����غ�ɫ����(MnO2),Լ10min����Һ�Ϻ�ɫ��dz |
�� | ȡ2mL0.1mol/LKMnO4��Һ���Թ��У������¿���1mLŨ��ˮ�����˰�ι�1:5�����ᣬ������Ƥ����ס�� | �����غ�ɫ����(MnO2)����Һ�Ϻ�ɫ���̱�dz��Լ5min����Һ�Ϻ�ɫ��ȫ��ȥ |
��3��ʵ��������������ΪN2��д���÷�Ӧ�����ӷ���ʽ:_________��
��4��ʵ���٢�˵��________________��
��5��ʵ������ʵ������Ӧ����_____(����������������)��ԭ����_________��
��6��1:5��������Һ(�ܶ�Ϊ��2g��cm-3)��������������Ϊ98%��Ũ����(�ܶ�Ϊ��1g��cm-3)��
����ˮ�������1:5��ɣ����1:5��������Һ�����ʵ���Ũ��Ϊ_____mol/L��(�ú���1����2��ʽ�ӱ�ʾ)
��7����ʵ��I��II�ɵó��Ľ�����____________________��