��Ŀ����
��ԴΣ����ʹ�������Ѱ���µ������Դ�Ϳ�չ��������Դ����Ч���á��ļ����з������ȼ�ϵ��ʹ��������ֱ�Ӳ��������DZ����������ս�ԵĿ���֮һ���Զ��飨��֪�����ȼ����Ϊ2 877.6 kJ��mol��1��Ϊ�����ش��������⣺
��1��д������ȼ�յ��Ȼ�ѧ����ʽ��________________��
��2���������ȼ���ȣ�2 878 kJ��mol��1���춡���ȼ���ȣ�2 869 kJ��mol����������ת��Ϊ�춡��Ĺ�����________����ų��������ա���������
��1��д������ȼ�յ��Ȼ�ѧ����ʽ��________________��
��2���������ȼ���ȣ�2 878 kJ��mol��1���춡���ȼ���ȣ�2 869 kJ��mol����������ת��Ϊ�춡��Ĺ�����________����ų��������ա���������
��1��2C4H10��g����13O2��g��=8CO2��g����10H2O��l������H����5 755.2 kJ��mol��1
��2���ų�
��2���ų�
��1��2C4H10��g����13O2��g��=8CO2��g����10H2O��l������H����5 755.2 kJ��mol��1����
2C4H10�������飬g����13O2��g��=8CO2��g����10H2O��l������H����5 756 kJ��mol��1����
2C4H10���춡�飬g����13O2��g��=8CO2��g����10H2O��l������H����5 738 kJ��mol��1����
���ø�˹���ɽ��ڣ����ٳ���2��C4H10�������飬g��=C4H10���춡�飬g������H����9 kJ��mol��1���÷�ӦΪ���ȷ�Ӧ��
2C4H10�������飬g����13O2��g��=8CO2��g����10H2O��l������H����5 756 kJ��mol��1����
2C4H10���춡�飬g����13O2��g��=8CO2��g����10H2O��l������H����5 738 kJ��mol��1����
���ø�˹���ɽ��ڣ����ٳ���2��C4H10�������飬g��=C4H10���춡�飬g������H����9 kJ��mol��1���÷�ӦΪ���ȷ�Ӧ��

��ϰ��ϵ�д�
 С��ſ�ʱ��ҵϵ�д�
С��ſ�ʱ��ҵϵ�д� һ������ϵ�д�
һ������ϵ�д� �Ƹ�С״Ԫ���ֳ������ϵ�д�
�Ƹ�С״Ԫ���ֳ������ϵ�д� �¸��̵�ѧϵ�д�
�¸��̵�ѧϵ�д� ����ͬѧһ����ʦȫ�źþ�ϵ�д�
����ͬѧһ����ʦȫ�źþ�ϵ�д�
�����Ŀ
 2PbSO4+2H2O
2PbSO4+2H2O CaO+CO2��
CaO+CO2��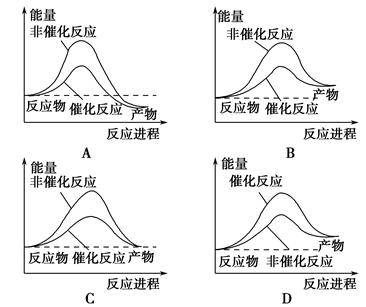
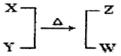
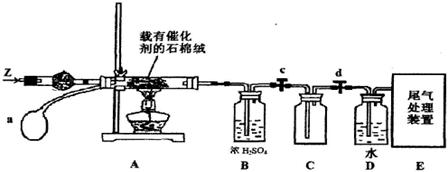
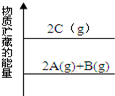
 2SO3Ϊ���ȷ�Ӧ�����ڸ÷�Ӧ������˵����ȷ����(����)��
2SO3Ϊ���ȷ�Ӧ�����ڸ÷�Ӧ������˵����ȷ����(����)��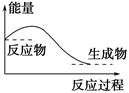
 2NH3(g)��H="-92.4" kJ/mol���÷�Ӧ�������仯��ͼ��ʾ��
2NH3(g)��H="-92.4" kJ/mol���÷�Ӧ�������仯��ͼ��ʾ��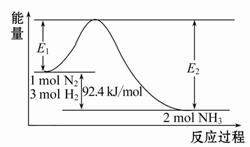
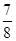 ����ʱH2��ת����Ϊ ������߸�������H2��ת���ʣ����д�ʩ���е��� (��ѡ����ĸ)��
����ʱH2��ת����Ϊ ������߸�������H2��ת���ʣ����д�ʩ���е��� (��ѡ����ĸ)��