��Ŀ����
14�������к�����ɳ��Ca2+��Mg2+��Fe3+��SO42-���ʣ�ijʵ��С�龫�ƴ���ˮ��ʵ��������£�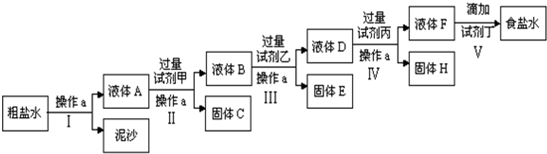
��ش��������⣺
��1������a���ò�������������������©�����ձ���
��2���ڢ��У���������Լ������������ִ��������ܳ��������Լ���Ϊ����������Һ���ѧ���ƻ�ѧʽ�����˲��з�����Ӧ�����ӷ���ʽ��Mg2++2OH-=Mg��OH��2����Fe3++3OH-=Fe��OH��3������д��һ�����Լ����Ļ�ѧʽHCl
��3���ɵõ���ʳ��ˮ��NaCl���˲�������Ϊ������
���� �����Dz�����ͨ�����˳�ȥ���������ʣ��õ���Һ�к�����������Ca2+��Mg2+��Fe3+��SO42-���ʣ���������Լ���Ϊ�������Ƴ��������Ӻ�þ���ӣ����˵õ���Һ�к���Ca2+��OH-��SO42-���ʣ���������Լ���Ϊ�Ȼ���������������ӣ����˵õ���Һ�к���OH-��Ca2+��Ba2+�������̼������Һ����ȥCa2+��Ba2+�����˵õ���Һ�к������������ӡ�̼������ӣ������������ᵽ��������������õ��ϴ�����ʳ��ˮ��
��1������a��ͨ�����˳�ȥ���������ʵIJ�����ʵ�ֹ������Һ����ķ����ǹ��ˣ����ݹ���װ�ûش���Ҫ��������
��2�����ݷ�����֪��Ϊ����������Һ�����ɵij���Ϊ����������������þ���ݴ�д����Ӧ�����ӷ���ʽ���Լ���Ϊ���
��3�����Ȼ�����Һ��ù����Ȼ��ƣ���Ҫͨ������������ɣ�
��� �⣺�����Dz�����ͨ�����˳�ȥ���������ʣ��õ���Һ�к�����������Ca2+��Mg2+��Fe3+��SO42-���ʣ���������Լ���Ϊ�������Ƴ��������Ӻ�þ���ӣ����˵õ���Һ�к���Ca2+��OH-��SO42-���ʣ���������Լ���Ϊ�Ȼ���������������ӣ����˵õ���Һ�к���OH-��Ca2+��Ba2+�������̼������Һ����ȥCa2+��Ba2+�����˵õ���Һ�к������������ӡ�̼������ӣ������������ᵽ��������������õ��ϴ�����ʳ��ˮ��
��1����ʳ��ˮ�У�ʵ����Һ����ɳ������ķ�����Բ��ù��˵ķ����������õ��������У���������©�����ձ���
�ʴ�Ϊ��©�����ձ���
��2�����ݷ�����֪��������Լ���ΪNaOH��Һ�������ķ�ӦΪ��Mg2++2OH-=Mg��OH��2����Fe3++3OH-=Fe��OH��3�����������Լ��������к��������������Ӻ�̼������ӣ��Ӷ��õ��������Ȼ�����Һ��
�ʴ�Ϊ���������ƣ�Mg2++2OH-=Mg��OH��2����Fe3++3OH-=Fe��OH��3����HCl��
��3���ɵõ���ʳ��ˮ��NaCl����Ҫ��ˮ������������������Ϊ�������ʴ�Ϊ��������
���� ������Ҫ������ε��ᴿ����Ŀ�Ѷ��еȣ����ճ��ӵķ�����ԭ��Ϊ���ؼ���ע���ȥ��������ʱ��Ҫ���Ǽ����Լ���˳��Ϊ�˱�֤�����ʳ����������Լ�������������Ϊ�˲������µ����ʣ����������Լ�Ҫ�ܹ���ȥǰ��������Ĺ����Լ���

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | ��ˮ�͵⻯�ط�Ӧ��Cl2+I-�TCl-+I2 | |
| B�� | ̼��������ᷴӦ��CO32-+2H+�TCO2��+H2O | |
| C�� | ������ˮ��Ӧ��Cl2+H2O�T2H++Cl-+ClO- | |
| D�� | ��ⱥ��ʳ��ˮ��2Cl-+2H2O$\frac{\underline{\;���\;}}{\;}$H2��+Cl2��+2OH- |
| A�� | O2 | B�� | Na2O | C�� | NaCl | D�� | SO2 |
| A�� | �����������к�ˮ���� | |
| B�� | �þƾ���ȡ��ˮ�еĵ� | |
| C�� | �������ķ�����ȥ������Һ�к��е�����NaCl | |
| D�� | �ü��ȡ������ķ������Գ�ȥ�����е�CaCl2��MgCl2������ |

| A�� | CH3CH2CH2CH3 | B�� | CH3CH2CH2OCH2CH2CH3 | ||
| C�� | ��CH3��2CH2OCH2��CH3��2 | D�� | CH3CH2OCH2CH3 |
| A�� | 40K��40Caԭ���е���������������������� | |
| B�� | H2��D2��Ϊͬλ�� | |
| C�� | ��Ϊͬλ�ص�ԭ�ӣ����ǵ�ԭ�Ӻ��ڵ�������һ������� | |
| D�� | ��Ϊͬλ�ص�ԭ�ӣ�����ԭ�Ӻ��ڵ�������һ����� |
| X | ||||
| Y | Z | W |
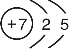 ��
����2��W��Z���⻯���У����ȶ�����HCl���û�ѧʽ��ʾ��
��3����Y��ij���γ�������ˮ�����侻ˮԭ����Al3+3H2O?Al��OH��3�����壩+3H+�������ӷ���ʽ��ʾ��
��������ѧ��ѧԭ��������������⣺
��4����֪��Si+2NaOH+H2O=Na2SiO3+2H2����ijͬѧ���õ��ʹ����Ϊ�缫�������ԭ��أ�NaOHΪ�������Һ������ԭ��ظ����ĵ缫��ӦʽΪSi+6OH--4e-=SiO32-+3H2O��
��5����֪����C��s��+O2��g��=CO2��g����H1��
��CO2��g��+C��s��=2CO��g����H2��
��Si��s��+O2��g��=SiO2��s����H3��
��ҵ�������ֹ���Ȼ�ѧ����ʽΪ2C��s��+SiO2��s��=Si��s��+2CO��g����H=��H1+��H2-��H3��
��6��25��ʱ����a mol•L-1NH3��ˮ��Һ�еμ�0.1mol•L-1H2SO4��Һ���μӹ�������Һ�ĵ���������ǿ�����ǿ���������������䡱����ʵ����a mol•L-1NH3��ˮ��Һ��pHֵΪ12����˰�ˮ�ĵ���ƽ�ⳣ��Kb=$\frac{1{0}^{-4}}{��a=1{0}^{-2}��}$mol•L-1���ú�a�Ĵ���ʽ��ʾ����