��Ŀ����
��ҵ���Ի�ͭ��Ϊԭ�ϣ����û��������������ָ֡�
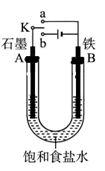
��1���ù��յ��м���̻ᷢ����Ӧ��2Cu2O+Cu2S 6Cu+SO2������Ӧ���������� ��
6Cu+SO2������Ӧ���������� ��
��2���������Ĵ�ͭ�����ʽ϶ࡣij��ѧ�о���ѧϰС����ʵ������������CuSO4��Һ�����Һ��ʵ�ִ�ͭ���ᴿ�����Ե�����Һ���о������Ӻͺ����ⶨ��
ʵ��һ ��ͭ���ᴿ��ͭ�к���������п������������Ƚ����������������ʣ������Ӧ�������ʱ��ͭӦ���Դ�� �������������ϵĵ缫��ӦʽΪ ��
ʵ��� ������Һ�ľ��������ھ���ͭ�Ĺ����У����Һ��c��Cu2+�����½���c��Fe2+����c��Zn2+���������������趨ʱ��ȥ���е�Fe2+��Zn2+����ͬѧ�ο��±������ݣ���������·�����
�Լ�a�� ���ѧʽ������Ŀ���� ���÷����ܹ���ȥ������������ �������ӷ��ţ���
ʵ���� ������Һ���Ӻ����IJⶨ
��ͬѧ��������·�����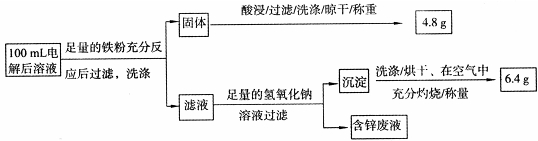
��100mL��Һ��Cu2+��Ũ��Ϊ mol��L-1��Fe2+��Ũ��Ϊ mol��L-1��
(14��)
��1��Cu2O ��Cu2S (2��)��
��2����(1��) Cu2++2e-= Cu(2��) H2O2(2��)��Fe2+������Fe3+(2��) Fe2+ (2��)
0.75(2��)0.05(2��)
�������������
��1����Ӧ��Cu2O ��Cu2S��Cu����+1��0�ۣ����Է�Ӧ��������Cu2O ��Cu2S��
��2������ͭʱ��ͭ���������Դ�������������缫��Ӧ��Cu2++2e-= Cu�����������е���pH��4��֪�������������Լ�a������������Fe2+������Fe3+����ȥFe2+������Ϊ����ͭ�Ļ����ᴦ�����4.8gΪͭ��n��Cu��=4.8/64g/mol=0.075mol��c��Cu2+��=0.75 mol��L-1��������������m��Fe2O3��=6.4g��n��Fe2O3��=6.4g/160g/mol=0.04mol����n��Fe��=0.08mol����������������ͭ���û���Ӧ��Ϊ0.075mol������ԭ��Һ��n��Fe��=0.005mol��Fe2+��Ũ��Ϊ0.05 mol��L-1��
���㣺����Ԫ�ؼ��仯����֪ʶ��������ԭ֪ʶ�����ء�ʵ���������������֪ʶ��

����16�֣������ͷ�ˮ���ؽ���Ԫ�ظ��Ķ��ԣ��ɽ�Cr2O72-ת��ΪCr(OH)3������ȥ�����ֽ����������ɳ�����pH�����
| | ��ʼ������pH | ��ȫ������pH |
| Fe(OH)2 | 7.0 | 9.0 |
| Fe(OH)3 | 1.9 | 3.2 |
| Cr(OH)3 | 6.0 | 8.0 |
��1��ij������ˮ��������Ҫ��������ͼ��ʾ��
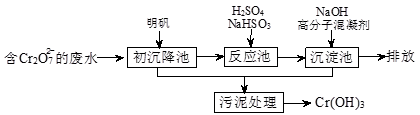
�ٳ������м����������������������õ�ԭ���ǣ������ӷ���ʽ��ʾ�� ��
���벹�䲢��ƽ���·�Ӧ���з�����Ҫ��Ӧ�����ӷ���ʽ��
Cr2O72-+ HSO3-+ == Cr3++ SO42-+ H2O��
�۸��ݡ����������͡��кͷ�����ԭ������������м���NaOH��Һ���˹����з�����Ҫ��Ӧ�����ӷ���ʽ�ǣ�H++OH-=H2O�� ��֤��Cr3+������ȫ�ķ����� ��
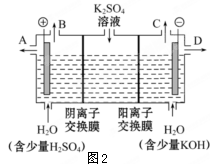
��2����ҵ���õ�ⷨ��������Cr2O72-��ˮ��ʵ����������ͼģ�����Cr2O72-�ķ�ˮ��������Ӧ��Fe-2e-=Fe2+��������Ӧʽ��2H++2e-=H2����
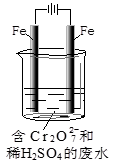
��Fe2+��������Һ�е�Cr2O72-��Ӧ�����ӷ���ʽ�� ��
��������Ӧ�õ��Ľ������������������ɳ�����ȫ�������ˮ�ĵ���ƽ��ǶȽ�����ԭ��
��
������Һ�к���0.01mol Cr2O72-����������ȫ�����ɳ��������ʵ�����_______mol��
���и�ʵ�飬�����ܳɹ����ǣ� ��
��������ͨ�ƾ�����ˮ�Ҵ��� �ڱ���Ũ�����ϼ�����60������������
���ñ�����ˮ������屽�� �����ѻ����ʹӵ�ˮ����ȡ��
| A��ֻ�Тٺ͢� | B��ֻ�Тں͢� | C��ֻ�Тٺ͢� | D���٢ڢۢ� |
�����й�ʵ���������ȷ��( )
| A����SO2ͨ����ˮ��Һ����ˮ��ɫ������˵��SO2��Ư���� |
| B�������ۺ�ϡ�����Ϻ���ȣ��ټ���������Һ���ȣ����Լ������ˮ���IJ��� |
| C����C2H5OH��Ũ����170�湲�ȣ��Ƶõ�����ֱ��ͨ������KMnO4��Һ�����Լ������ɵ���ϩ���� |
| D����AgCl��Һ�еμ�KI��Һ����ɫ����ת��Ϊ��ɫ����������˵��AgI��AgCl������ |
��ȥ���������������Լ��ͷ�������ȷ����
| A���Ҵ������ᣩ�� KOH��Һ ����Һ |
| B����ϩ��SO2�� �� KMnO4��Һ ��ϴ�� |
| C�����飨��ϩ���� H2���������������� |
| D���Ҵ���ˮ���� CaO ������ |
ij�����л��һ��������̼ԭ������������10�����Ľṹ��ʽ�ɱ�ʾΪ ����1H�˴Ź�����ͼ�г�������壬�Ҳ�ͬ��ѧ��������ԭ����֮��Ϊ3��2�����й��ڸ��л����˵���������
����1H�˴Ź�����ͼ�г�������壬�Ҳ�ͬ��ѧ��������ԭ����֮��Ϊ3��2�����й��ڸ��л����˵���������
| A�����л��ﲻ����Ϊ���㴼 | B�����л���ķ���ʽ����ΪC8H10O2 |
| C�����л��������NaOH��Һ��Ӧ | D�����л���ȼ��ʱһ��ֻ����CO2��H2O |