��Ŀ����
����Ŀ���������һ�����ᣬ��������ʴ��������֪25��ʱ��
HF(aq) + OH��(aq)![]() F��(aq) + H2O(l) ��H=��67.7 kJ/mol
F��(aq) + H2O(l) ��H=��67.7 kJ/mol
H+(aq) + OH��(aq)![]() H2O(l) ��H=��57.3kJ/mol
H2O(l) ��H=��57.3kJ/mol
��10 mL 0.1mol/L��NaOH��Һ�У�����10 mL Ũ��Ϊc mol/L��HFϡ��Һ������˵���д������
A. ���������Һ�¶����ߣ�HF�ĵ���̶ȼ�С�������ǻӷ���
B. ˮ������Ȼ�ѧ����ʽΪ��H2O(1)![]() H+(aq)+OH��(aq)����H= +57.3kJ/mol
H+(aq)+OH��(aq)����H= +57.3kJ/mol
C. ��c > 0.1ʱ��һ�������ڣ�c(Na+) = c(F��)
D. ����Ϻ���Һ�У�c(Na+)>c(OH��) >c(F��)>c(H+)����cһ��С��0.1
���𰸡�C
�����������������C���ΪHF�����ᣬ������ʣ����ֵ��룬��F����ˮ��̶ȵ��ڶ����HF�ĵ���̶�ʱ�����Գ���c(Na+)=c(F��)���ʴ�����ѡC��

 ��ѧ��������������Ͼ���ѧ������ϵ�д�
��ѧ��������������Ͼ���ѧ������ϵ�д�����Ŀ������ʵ��������ʵ����ۻ�ԭ����һ�µ���
ѡ�� | ʵ������ | ʵ����ۻ�ԭ�� |
A | ����������ĭ�������� | 3HCO3-+Al3+===Al(OH)3��+3CO2�� |
B | ��AgCl����Һ�е�������KI��Һ���л�ɫ�������� | ˵��KSP��AgCl����KSP��AgI�� |
C | ��NaHS��Һ�е����̪����Һ���ɫ | HS-ˮ��̶ȴ��ڵ���̶� |
D | Na2CO3��Һ�еμӷ�̪�ʺ�ɫ | CO32-+2H2O |
A. A B. B C. C D. D
����Ŀ��SO2�㷺����ҽҩ�����Ṥҵ�������շ����е�SO2�������·�����
������ | �ü�ʽ������Al2(SO4)x(OH)y��Һ���ո���SO2 |
������ | ��Fe2+��Fe3+���£��ÿ���(O2)��SO2����ΪH2SO4 |
��1��������Ĺ������¡�
�� �Ʊ�Al2(SO4)x(OH)y
��Al2(SO4)3��Һ�м���CaO��ĩ����pH��3.6�� CaO������֮һ�Ǵٽ�_______ˮ�⣨�����ӷ��ţ���ͬ�������dz���һ����_______��
�� ���գ�Al2(SO4)x(OH)y����SO2��IJ�����_______��д��ѧʽ����
�� ���������Ȣ��в������SO2��Al2(SO4)x(OH)y������
��2���������У���Fe2+���£�SO2��O2��H2O����H2SO4�Ļ�ѧ����ʽ��______��
��3���������У�Fe2+�Ĵ����̿ɱ�ʾ���£�
����2 Fe2++ SO2+ O2=2 Fe3++ SO42-
���� ����
�� д���������ӷ���ʽ��______��
�� ����ʵ�鷽����֤ʵ���������̡���ʵ�鷽������������
a����FeCl2��Һ����KSCN���ޱ仯
b����FeCl2��Һͨ������SO2������KSCN����ɫ��졣
c��ȡb����Һ��_______��
��4���������У����������õζ����ⶨ�����в���SO2�ĺ�������V L���ѻ���Ϊ��״���������е�SO2��1%��H2O2��ȫ���գ�����Һ����ͼ��ʾװ�õζ���������a mL c mol/L NaOH��Һ��

�� H2O2����SO2�Ļ�ѧ����ʽ_______��
�� �����в���SO2���������Ϊ_______��
����Ŀ��ijС����Ʋ�ͬʵ�鷽���Ƚ�Cu2+��Ag+ �������ԡ�
��1������1��ͨ���û���Ӧ�Ƚ�
���ữ��AgNO3��Һ����ͭ˿��������ɫ���壬��Һ��������Ӧ�����ӷ���ʽ��_______��˵��������Ag+��Cu2+��
��2������2��ͨ��Cu2+��Ag+ �ֱ���ͬһ���ʷ�Ӧ���бȽ�
ʵ�� | �Լ� | ��ż����� | |
�Թ� | �ι� | ||
| 1.0 mol/L KI��Һ | 1.0 mol/L AgNO3��Һ | ������ɫ��������Һ��ɫ |
1.0 mol/L CuSO4��Һ | ������ɫ����A����Һ��� | ||
�� �����飬������Һ����I2����ɫ������________��
�� �����飬������Һ��I2���Ʋ�Cu2+������������ɫ����A��CuI��ȷ��A��ʵ�����£�

a��������Һ��I2����Һ����ɫ˵����Һ����________�������ӷ��ţ���
b����ɫ����B��________��
c����ɫ����A��AgNO3��Һ��Ӧ�����ӷ���ʽ��____��˵��������Ag+��Cu2+��
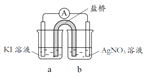
��3����������2��Ag+ δ������I- ����Cu2+������I-��ԭ�����ʵ�����£�
��� | ʵ��1 | ʵ��2 | ʵ��3 |
ʵ�� |
|
|
|
���� | �����Ա仯 | a����Һ�Ͽ���ػ�ɫ,b�е缫 ����������������ָ��ƫת | c����Һ������dz��ɫ�� ������ָ��ƫת |
���缫��Ϊʯī����ҺŨ�Ⱦ�Ϊ 1 mol/L��b��d����ҺpH��4��
�� a����Һ���ػ�ɫ��ԭ����_______���õ缫��Ӧʽ��ʾ����
�� ��ʵ��3������˵��Cu2+������I-�������ǿ����е�����Ҳ���������ã����ʵ��֤ʵ�˸����ݣ�ʵ�鷽����������_______��
�� ����2�У�Cu2+������I-,��Ag+δ������I-��ԭ��_______��
�����ϣ�Ag+ + I- = AgI�� K1 =1.2��1016��2Ag+ + 2I- = 2Ag��+ I2 K2 = 8.7��108��