��Ŀ����
7��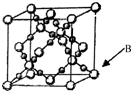 ijԭ�Ͼ��ⶨ��Ҫ����A��B��C��D��E����ǰ������Ԫ�أ���ԭ��������������Ԫ��A��B��C��D��E��ԭ�ӽṹ����Ϣ���£�
ijԭ�Ͼ��ⶨ��Ҫ����A��B��C��D��E����ǰ������Ԫ�أ���ԭ��������������Ԫ��A��B��C��D��E��ԭ�ӽṹ����Ϣ���£�| Ԫ�� | Ԫ�����ʻ�ԭ�ӽṹ |
| A | ���ڱ���ԭ�Ӱ뾶��С |
| B | ԭ�Ӻ��������ֲ�ͬ��������ԭ�ӹ���Ҹ�����������ĵ�������ͬ |
| C | �����p�������� |
| D | λ�ڶ����ڣ���ԭ�ӵĺ���ɶԵ�������δ�ɶԵ�������3�� |
| E | λ��ds����ԭ�ӵ�������������A����ͬ |
��1��B��C��D��һ��������С����ΪN��O��C��
��2��E�Ķ������ӵĵ����Ų�ʽΪ1s22s22p63s23p63d9��
��3��A2B2D4��һ���л����ᣬ��������������÷�����B���ӻ���ʽΪsp2��1mol A2B2D4�����к��ЦҼ���ĿΪ7��6.02��1023��
��4���뻯����BD��Ϊ�ȵ�����������ӻ�ѧʽΪCN-�ȣ���дһ�֣���
��5��B2A6��C2A4�����о�����18�����ӣ����ǵķе����ϴ���Ҫԭ����N2H4����֮����������
��6��BD2�ڸ��¸�ѹ�����γɾ���ľ�������ͼ��ʾ��һ���þ����к�16��Dԭ�ӣ�
���� A��B��C��D��E����ǰ������Ԫ�أ���ԭ�����������������ڱ���AԪ��ԭ�Ӱ뾶��С����AΪHԪ�أ�BԪ��ԭ�Ӻ��������ֲ�ͬ���ܼ��Ҹ����ܼ������ĵ�������ͬ����������Ų�Ϊ1s22s22p2����BΪ̼Ԫ�أ�Dλ�ڶ����ڣ���ԭ�ӵĺ���ɶԵ�������δ�ɶԵ�������3��������������ԭ�ӵ����Ӵ���p�ܼ������3��������ɶԵ�������ֻ��Ϊ6����������Ϊ2����������Ų�Ϊ1s22s22p4����DΪOԪ�أ�CԪ��ԭ�������P����������ԭ������С�����������������Ų�Ϊ1s22s22p3����BΪNԪ�أ�Eλ��ds����ԭ�ӵ�������������A����ͬ����Χ�����Ų�Ϊ3d104s1����EΪCu���ݴ˽��
��� �⣺A��B��C��D��E����ǰ������Ԫ�أ���ԭ�����������������ڱ���AԪ��ԭ�Ӱ뾶��С����AΪHԪ�أ�BԪ��ԭ�Ӻ��������ֲ�ͬ���ܼ��Ҹ����ܼ������ĵ�������ͬ����������Ų�Ϊ1s22s22p2����BΪ̼Ԫ�أ�Dλ�ڶ����ڣ���ԭ�ӵĺ���ɶԵ�������δ�ɶԵ�������3��������������ԭ�ӵ����Ӵ���p�ܼ������3��������ɶԵ�������ֻ��Ϊ6����������Ϊ2����������Ų�Ϊ1s22s22p4����DΪOԪ�أ�CԪ��ԭ�������P����������ԭ������С�����������������Ų�Ϊ1s22s22p3����BΪNԪ�أ�Eλ��ds����ԭ�ӵ�������������A����ͬ����Χ�����Ų�Ϊ3d104s1����EΪCu��
��1��ͬ������ԭ����������һ�����ܳ��������ƣ���NԪ��2p�ܼ�����3�����ӣ�Ϊ�����ȶ�״̬�������ϵͣ���һ�����ܸ���ͬ��������Ԫ�أ��ʵ�һ������N��O��C��
�ʴ�Ϊ��N��O��C��
��2��EΪCuԪ�أ�Cu2+�ĵ����Ų�ʽΪ1s22s22p63s23p63d9��
�ʴ�Ϊ��1s22s22p63s23p63d9��
��3��H2C2O4��������������ṹ��ʽΪHOOC-COOH��������Cԭ���γ�3���Ҽ���û�йµ��Ӷԣ��÷�����̼ԭ�ӵ��ӻ���ʽΪsp2������Ϊ�Ҽ���˫������1���Ҽ���1���м�����HOOC-COOH�����к���7���Ҽ���1molA2B2D4�����к��ЦҼ���ĿΪ7��6.02��1023��
�ʴ�Ϊ��sp2��7��6.02��1023��
��4��ԭ��������ͬ���۵���������ͬ������Ϊ�ȵ����壬�뻯����CO��Ϊ�ȵ�����������ӻ�ѧʽΪ CN-�ȣ�
�ʴ�Ϊ��CN-�ȣ�
��5��C2H6��N2H4�����о�����18�����ӣ����ǵķе����ϴ���Ҫԭ���ǣ�N2H4����֮����������
�ʴ�Ϊ��N2H4����֮����������
��6��CO2�ڸ��¸�ѹ�����γɾ���ľ����к�ɫ��ΪOԭ�ӣ���Ϊ�����ڲ���һ���þ����к�Oԭ����ĿΪ16��
�ʴ�Ϊ��16��
���� �����Ƕ����ʽṹ�Ŀ��飬�漰�����ܡ���������Ų�����ѧ�����ӻ����������������ṹ�ȣ�ע����������ͬ���ڵ�һ�������쳣ԭ���Ѷ��еȣ�

 ��ѧ�̸̳����¿α�ϵ�д�
��ѧ�̸̳����¿α�ϵ�д� Сѧ��ʱ��ѵϵ�д�
Сѧ��ʱ��ѵϵ�д�| A�� | 18g D2O��18g H2O�к��е���������Ϊ10NA | |
| B�� | 2L 0.5 mol•L-1��������Һ�к��е�H+������Ϊ2NA | |
| C�� | ����������ˮ��Ӧʱ������0.1mol����ת�Ƶĵ�����Ϊ0.2NA | |
| D�� | �ܱ�������2mol NO��1mol O2��ַ�Ӧ������ķ�����Ϊ2NA |
| ѡ�� | ���� | ��; | ���� |
| A | H2O2 | ʳƷƯ�� | H2O2����ǿ�����ԣ���Ư��ʳƷ |
| B | SiO2 | �����оƬ | SiO2��һ�����õİ뵼�� |
| C | Fe3O4 | ��ɫͿ�� | Fe3O4��һ�ֺ�ɫ������ |
| D | ŨH2SO4 | ����� | ŨH2SO4����ˮ�� |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | ϡ���� | B�� | ϡ���� | C�� | ����ͭ | D�� | �������� |
| ѡ�� | �������ʵ | ���� |
| A | ���ȵĴ�����Һϴȥ���� | Na2CO3��ֱ�Ӻ����۷�Ӧ |
| B | Ư���ڿ����о��ñ��� | Ư���е�CaCl2������е�CO2��Ӧ����CaCO3 |
| C | ʩ��ʱ����ľ�ң���Ч�ɷ�ΪK2CO3��������NH4Cl���ʹ�� | K2CO3��NH4Cl�������з�Ӧ���ɰ����ή�ͷ�Ч |
| D | �����ڳ�ʪ�Ŀ������������� | �����ڿ����з��������ⸯʴ |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | Al2O3�����ͻ���ϡ�Al��OH��3������ȼ�� | |
| B�� | ̼�ظֵ���Ҫ�ɷ�����̼�Ͻ�˾ĸ�춦����Ҫ�ɷ���ͭ���Ͻ� | |
| C�� | ��Ȼ���в���������̬�Ĺ裬����Ҫ�Զ�����������ε���ʽ���� | |
| D�� | ͨ����ѧ�仯����ʵ��12C��14C����ת�� |
| A�� | BaSO4�TBa2++SO42- | |
| B�� | Na2CO3�TNa++CO32- | |
| C�� | SO3����ˮ��Һ���Ե��磬�������ڵ���� | |
| D�� | CaCO3Ͷ��ˮ�У���Һ�����磬˵��CaCO3�Ƿǵ���� |
| ʵ����� | ���� | ���ͻ���� | |
| A | ���������CH4��Cl2װ��Ӳ�ʴ��Թܻ�Ϻ��ٽ��Թܵ����ڱ���ʳ��ˮ�У��������������³�ַ�Ӧ | �Թ���������ɫ��dz���Թ��ڱڳ�����״Һ�� | �����������ڹ��������·�����ȡ����Ӧ |
| B | ��ʢ����ˮ�����Թ��зֱ����һ��������ϩ�ͱ� | ���Թ��е�ˮ�㶼����ɫ | ��ϩ�ͱ�������ˮ�����˼ӳɷ�Ӧ |
| C | ��ʢ��������ˮ�Ҵ����Թ��У�����һС�����еġ�����ֽ���ɱ���ú�͵Ľ����� | �Ƹ�����ˮ�Ҵ����棬��Ӧ������ˮ�и����� | �Ҵ��ṹ���ǻ���Ļ��Ա�ˮ�е�ǿ |
| D | ȡ1mL 20%��������Һ������3��5��ϡ���ᣮˮԡ����5min��ȡ������Һ��ֱ�Ӽ�����������Cu��OH��2������4min | δ����ש��ɫ���� | ���Dz���ˮ������������ |
| A�� | A | B�� | B | C�� | C | D�� | D |