��Ŀ����
һ���¶��£�lmol TaI4��n mol S2��2L���ܱ������з�����ӦTaI4��g��+S2��g��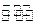 2TaS2��s��+2I2��g����t min��ﵽƽ�⣬��ʱ����2a mol I2������˵����ȷ���� �� ��
2TaS2��s��+2I2��g����t min��ﵽƽ�⣬��ʱ����2a mol I2������˵����ȷ���� �� ��
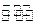 2TaS2��s��+2I2��g����t min��ﵽƽ�⣬��ʱ����2a mol I2������˵����ȷ���� �� ��
2TaS2��s��+2I2��g����t min��ﵽƽ�⣬��ʱ����2a mol I2������˵����ȷ���� �� ��A����TaI4��ʾ�˷�Ӧ�ķ�Ӧ������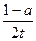 mol����L��min�� mol����L��min�� |
| B����ƽ������ϵ�м���lmol TaS2��ƽ�����淴Ӧ�����ƶ� |
| C����ƽ������ϵ���£���ѧƽ��һ���ƶ� |
| D����ƽ������ϵ�г���lmol TaI4��v����������v���棩��С��ƽ�������ƶ� |
C
��

��ϰ��ϵ�д�
�����Ŀ
 2NH3(g)����һ���¶��£���һ������N2��H2ͨ��̶����Ϊ1L���ܱ������дﵽƽ��ı�������������ʹƽ��������Ӧ�����ƶ����� ��
2NH3(g)����һ���¶��£���һ������N2��H2ͨ��̶����Ϊ1L���ܱ������дﵽƽ��ı�������������ʹƽ��������Ӧ�����ƶ����� ��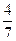 ������������´ﵽƽ��ʱN2ת����Ϊ ��
������������´ﵽƽ��ʱN2ת����Ϊ ��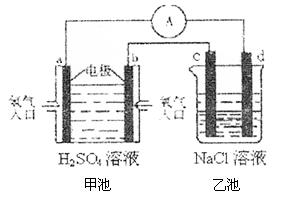

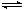 2NH3����ƽ���÷�Ӧ�ų�������Ϊ18.4 kJ����������ܵ����ʵ���Ϊ3.6 mol�������ڵ�ѹǿ��Ϊԭ����90%����֪N2��H2��ת������ͬ��
2NH3����ƽ���÷�Ӧ�ų�������Ϊ18.4 kJ����������ܵ����ʵ���Ϊ3.6 mol�������ڵ�ѹǿ��Ϊԭ����90%����֪N2��H2��ת������ͬ�� ��
�� ��ƽ����ȫ��ͬ��
��ƽ����ȫ��ͬ�� 0�֣�830Kʱ�����ܱ������з������п��淴Ӧ��CO��g��+H2O��g��
0�֣�830Kʱ�����ܱ������з������п��淴Ӧ��CO��g��+H2O��g�� CO2��g��+H2��g�� ��H��0 ���Իش��������⣺
CO2��g��+H2��g�� ��H��0 ���Իش��������⣺ -1���ﵽƽ��ʱCO��ת����Ϊ60%�����ڴ��¶��£��÷�Ӧ��ƽ�ⳣ��K=
-1���ﵽƽ��ʱCO��ת����Ϊ60%�����ڴ��¶��£��÷�Ӧ��ƽ�ⳣ��K=  ���ڡ�����С�ڡ����ڡ�����
���ڡ�����С�ڡ����ڡ����� ����С�����䡱����
����С�����䡱���� 2SO3���ڻ�����г���һ������18Oԭ�ӣ��ﵽƽ���Ժ�18Oԭ��
2SO3���ڻ�����г���һ������18Oԭ�ӣ��ﵽƽ���Ժ�18Oԭ�� O3��
O3��
 2C B��A��B
2C B��A��B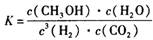 ���¶�����,Kֵ��С������˵����ȷ����
���¶�����,Kֵ��С������˵����ȷ����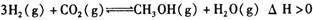
 CH3OH��g����T1��T2�¶��£�CH3OH�����ʵ�������ʱ��ı仯��ϵ����ͼ��ʾ�������й�˵����ȷ����
CH3OH��g����T1��T2�¶��£�CH3OH�����ʵ�������ʱ��ı仯��ϵ����ͼ��ʾ�������й�˵����ȷ����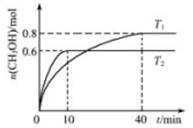
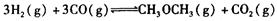
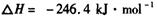
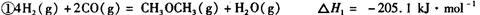

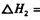 ��__________________ ______________
��__________________ ______________ 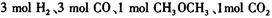 ����һ���¶Ⱥ�ѹǿ�·�����Ӧ��
����һ���¶Ⱥ�ѹǿ�·�����Ӧ��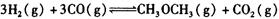 ����һ��ʱ��ﵽƽ�⣬�����ƽ��ʱ��������ܶ���ͬ��ͬѹ����ʼʱ��1.6�����ʣ��ٷ�Ӧ��ʼʱ�����淴Ӧ����
����һ��ʱ��ﵽƽ�⣬�����ƽ��ʱ��������ܶ���ͬ��ͬѹ����ʼʱ��1.6�����ʣ��ٷ�Ӧ��ʼʱ�����淴Ӧ���� �Ĵ�С��V(����____V���棩��� >������ < ����"="),�����ǣ�________________________________
�Ĵ�С��V(����____V���棩��� >������ < ����"="),�����ǣ�________________________________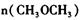 =_________________��ƽ��ʱCO��ת����= _________________��
=_________________��ƽ��ʱCO��ת����= _________________��