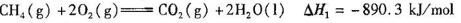��Ŀ����
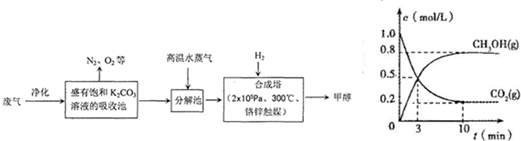
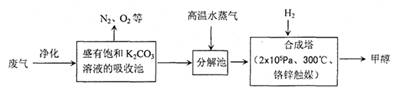
��16�֣�Ϊ���������CO2�ĺ�����������⣬ij��ѧ���������ɫ���ɡ����룺�ѹ����ų��ĸ���CO2�ķ�������������̼�����Һ���գ�Ȼ���ٰ�CO2����Һ����ȡ����������ѧ��Ӧʹ�����е�CO2ת��Ϊȼ�ϼ״�������ɫ���ɡ�����IJ��ּ����������£�
�ϳ����з�Ӧ�Ļ�ѧ����ʽΪ ����H<0����ƽ���ƶ�ԭ���������������������ԭ������ƽ��ת���ʡ���ʵ�������в���300����¶ȣ��������¶ȶԷ�Ӧ���ʵ�Ӱ���⣬����Ҫ������ ��
�Ӻϳ���������״���ԭ�������� ������ԭ���Ƚ����������ĸ����
A������ B����Һ C������ D���ᾧ
��3����ҵ������һ��������ѭ�����á�����ѭ�����á������Ч�桢���ܻ�������Ҫ��ʩ������ɫ���ɡ����뼼���������ܹ���ѭ�����á��ģ���K2CO3��Һ��CO2��H2�⣬������ ��
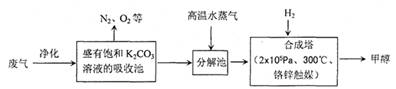
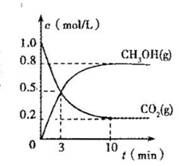
��4�������Ϊ2L�ĺϳ����У�����2 mol CO2��6 mol H2�����CO2(g)��CH3OH(g)��Ũ����ʱ��仯��ͼ��ʾ���ӷ�Ӧ��ʼ��ƽ�⣬v��H2) =______________����ʹƽ����ϵ��n(CH3OH)/n(CO2)����Ĵ�ʩ�� ��
��5���罫CO2��H2��1:4������Ȼ�ϣ����ʵ��������¿��Ƶ�CH4��
��֪CH4(g)+2O2(g)= CO2(g)+2H2O(l) ��H1����890.3kJ/mol
H2 (g)+ O2(g)= H2O(l) ��H2����285.8kJ/mol
д��CO2(g)��H2(g)��Ӧ����CH4(g)��Һ̬ˮ���Ȼ�ѧ����ʽ________________��
��16�֣�
��1�� CO2+3H2�� CH3OH+H2O ��3�֣�;�������Ĵ����ԣ�2 �֣�
��2��C��2�֣�
��3������ˮ������2�֣���
��4��0.24mol/(L��min)��2�֣�������H2�������ȣ�2�֣�
��5��CO2(g)+4H2 (g) = CH4(g)+2H2O(l) ��H����252.9kJ/mol��3�֣�
����������

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�