��Ŀ����
��1���£�N2H4���ֳ���������һ�ֿ�ȼ�Ե�Һ�壬���������ȼ�ϡ���֪��101kPaʱ��3.20gN2H4����������ȫȼ�����ɵ������ų�����62.4kJ��25��ʱ����N2H4��ȫȼ�շ�Ӧ���Ȼ�ѧ����ʽ�� ��
��2���¡�����ȼ�ϵ����һ�ּ���ȼ�ϵ�أ��������Һ��20%~30%��KOH��Һ���¡�����ȼ�ϵ�طŵ�ʱ�������ĵ缫��Ӧʽ�� �������ĵ缫��Ӧʽ�� _ _��
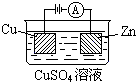
��3����ͼ��һ���绯ѧ����ʾ��ͼ��

��пƬ�Ϸ����ĵ缫��Ӧ�� ��
�ڼ���ʹ���¡�����ȼ�ϵ����Ϊ�������еĵ�Դ��ͭƬ�������仯128g������һ����ȼ�ϵ�����������ı��״���µĿ��� L����������������������Ϊ20%����
��4����ͳ�Ʊ��µķ�������NaClO����NH3���Ƶ��µ�ϡ��Һ���÷�Ӧ�����ӷ���ʽ�� ��
��1��N2H4(1)+ O2(g)=N2(g)+2H2O��1������H=��624kJ/mol��3�֣�
��2��O2+2H2O+4e��=4OH�� ����2�֣� N2H4+4OH����4e��=4H2O+N2����2�֣�
��3����Cu2++2e��=Cu ��2�֣� ��112��2�֣�
��4��ClO��+2NH3=N2H4+Cl��+H2O��2�֣�
����������

 ����С״Ԫ��������������ϵ�д�
����С״Ԫ��������������ϵ�д� ����������ϵ�д�
����������ϵ�д�
 ��1���£�N2H4���ֳ���������һ�ֿ�ȼ�Ե�Һ�壬���������ȼ�ϣ���֪��101kPaʱ��32.0gN2H4����������ȫȼ�����ɵ������ų�����624kJ��25��ʱ����N2H4��ȫȼ�շ�Ӧ���Ȼ�ѧ����ʽ��
��1���£�N2H4���ֳ���������һ�ֿ�ȼ�Ե�Һ�壬���������ȼ�ϣ���֪��101kPaʱ��32.0gN2H4����������ȫȼ�����ɵ������ų�����624kJ��25��ʱ����N2H4��ȫȼ�շ�Ӧ���Ȼ�ѧ����ʽ��