��Ŀ����
����Ŀ��KAl(SO4)2��12H2O����������һ�ָ���������ֽ�ȷ���Ӧ�ù㷺��ʵ�����У����÷������ޣ���Ҫ�ɷ�ΪAl������������Fe��Mg���ʣ��Ʊ������Ĺ�������ͼ��ʾ���ش��������⣺
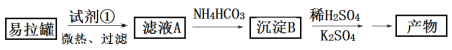
��1��Ϊ�������������ʣ��Լ���Ӧѡ��___�����ţ���
a��HCl��Һ b��H2SO4��Һ c����ˮ d��NaOH��Һ
��2���������ܽ��������Ҫ��Ӧ�Ļ�ѧ����ʽΪ________��
��3������B�Ļ�ѧʽΪ________����������������ˮ����Һ�������ԣ���ԭ����__________��
��4����֪��Kw=1.0��10-14��Al(OH)3![]() AlO2-+H++H2O K=2.0��10-13��Al(OH)3����NaOH��Һ��Ӧ��ƽ�ⳣ������_________��
AlO2-+H++H2O K=2.0��10-13��Al(OH)3����NaOH��Һ��Ӧ��ƽ�ⳣ������_________��
���𰸡���8�֣�
��1��d ��2�֣�
��2��2Al+2NaOH+2H2O=2NaAlO2+3H2�� ��2�֣�
��3��Al(OH)3 Al3+ˮ�⣬ʹ��Һ��H+Ũ������ ��ÿ��1�֣���2�֣�
��4��20 ��2�֣�
����������1�������ޣ���Ҫ�ɷ�ΪAl������������Fe��Mg���ʣ������Ƕ��DZȽϻ��õĽ��������������ᷢ����Ӧ����Al������ǿ����Һ��Ӧ��ΪNaAlO2������Һ����Fe��Mg����������Ӧ������Ҫ�Ժ�Al��������Ϊԭ����ȡ������Ӧ����ǿ��NaOH��Һ�ܽ⣬ ѡ��d��ȷ��Mg��Fe���ܽ⣬�����������У���2���������ܽ��������Ҫ��Ӧ�Ļ�ѧ����ʽΪ2Al+2NaOH+2H2O==2NaAlO2+3H2������Ӧ�����ӷ���ʽ��2Al+2OH-+2H2O=2AlO2-+3H2������3����ҺA����Ҫ�ɷ���NaAlO2������ˮ��Һ�м���NH4HCO3��Һ��AlO2-��NH4+�����ε�˫ˮ�ⷴӦ������Al(OH)3������NH3��H2O����Ӧ�ķ���ʽ��AlO2-+2H2O+NH4+= Al(OH)3��+NH3��H2O�����Գ���B��ѧʽΪAl(OH)3����������������ˮ����Һ�������ԣ���ԭ���Ǹ�����ǿ�������Σ�Al3+����ˮ�ⷴӦ������ˮ���������OH-�������մﵽƽ��ʱ��Һ��c(H+)>c(OH-)��������Һ�����ԡ���4����֪��Kw=1.0��10-14��Al(OH)3![]() AlO2-+H++H2O K=2.0��10-13��Al(OH)3����NaOH��Һ��Ӧ��ƽ�ⳣ��K=
AlO2-+H++H2O K=2.0��10-13��Al(OH)3����NaOH��Һ��Ӧ��ƽ�ⳣ��K=![]() ��
��








