��Ŀ����
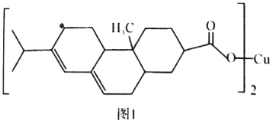
����Ŀ���ɻ���Ӧ���л��ϳ��о�����ҪӦ�ã�ij��״������G�ĺϳɹ������£�
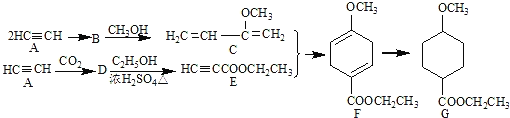
(1)A��BΪ�ӳɷ�Ӧ����B�Ľṹ��ʽ��______��B��C�ķ�Ӧ������______��
(2)G�к��еĹ�����������______��F�Ļ�ѧʽΪ______��
(3)D��E�Ļ�ѧ����ʽ��___________________________________________��
(4)H��F��ͬ���칹�壬�������нṹ�������ٺ˴Ź������׳��������շ������1�����շ壻�ڴ��ڼ�������CH3O������H�Ľṹ��ʽ��________________��
(5)��Cͨ���Ӿ۷�Ӧ�ϳɵĸ߷��ӻ�����M�Ļ�ѧ����ʽΪ______��
(6)����˵����ȷ����______��
A��A�ܺ�HCl��Ӧ�õ�������ϩ�ĵ���
B��1mol G��ȫȼ������8mol H2O
C��F�ܷ����ӳɷ�Ӧ��ȡ����Ӧ��������Ӧ��
���𰸡�HC��C��CH=CH2�ӳɷ�Ӧ�Ѽ�������C10H14O3HC��CCOOH+CH3CH2OH![]() HC��CCOOCH2CH3+H2O
HC��CCOOCH2CH3+H2O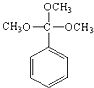
 AC
AC
��������
��1���Ƚ�A��C�Ľṹ������A��BΪ�ӳɷ�Ӧ������֪��BΪHC��C��CH=CH2���Ƚ�B��C�Ľṹ����֪����B��C�ķ�Ӧ�����Ǽӳɷ�Ӧ��
��2������G�Ľṹ��ʽ��֪G�к��еĹ���������������F�Ľṹ��ʽȷ��F�Ļ�ѧʽ��
��3���Ƚ�A��E�Ľṹ��ʽ����֪������Ȳ�������̼��Ӧ����DΪHC![]() CCOOH��HC
CCOOH��HC![]() CCOOH���Ҵ�����������Ӧ��E��
CCOOH���Ҵ�����������Ӧ��E��
��4���������������˴Ź������׳��������շ������1�����շ���˵�������ϵIJ�����һ���Գƽṹ�������ڼ�������CH3O���������F�Ľṹȷ��H�Ľṹ��ʽ��
��5������C�Ľṹ��ʽ����ϼӾ۷�Ӧ���ص��д��C�����Ӿ۷�Ӧ�ϳɵĸ߷��ӻ�����M�Ļ�ѧ����ʽ��
��6��A.��Ȳ��HCl���е����ʵ����ļӳɷ�Ӧ�õ�����ϩ��
B.����G�Ľṹ��ʽ����֪����G��������18����ԭ��������1mol G��ȫȼ������9mol H2O��
C.����F�Ľṹ��ʽ����֪����F����̼̼˫����������������F�ܷ����ӳɷ�Ӧ��ȡ����Ӧ��������Ӧ���ݴ˴�����
��1���Ƚ�A��C�Ľṹ������A��BΪ�ӳɷ�Ӧ������֪��BΪHC��C��CH=CH2���Ƚ�B��C�Ľṹ����֪����B��C�ķ�Ӧ�����Ǽӳɷ�Ӧ��
��ˣ�������ȷ������HC��C��CH=CH2���ӳɷ�Ӧ��
��2������G�Ľṹ��ʽ��֪G�к��еĹ���������Ϊ�Ѽ�������������F�Ľṹ��ʽ����֪��F�Ļ�ѧʽΪC10H14O3��
��ˣ�������ȷ�������Ѽ���������C10H14O3��
��3���Ƚ�A��E�Ľṹ��ʽ����֪������Ȳ�������̼��Ӧ����DΪHC![]() CCOOH��HC
CCOOH��HC![]() CCOOH���Ҵ�����������Ӧ��E��
CCOOH���Ҵ�����������Ӧ��E��
��Ӧ����ʽΪHC��CCOOH+CH3CH2OH![]() HC��CCOOCH2CH3+H2O��
HC��CCOOCH2CH3+H2O��
��ˣ�������ȷ������HC��CCOOH+CH3CH2OH![]() HC��CCOOCH2CH3+H2O��
HC��CCOOCH2CH3+H2O��
��4���������������˴Ź������׳��������շ������1�����շ���˵�������ϵIJ�����һ���Գƽṹ�������ڼ�������CH3O���������F�Ľṹ����֪��H�Ľṹ��ʽΪ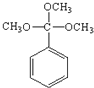 ��
��
��ˣ�������ȷ������ ��
��
��5������C�Ľṹ��ʽ����֪����C�����Ӿ۷�Ӧ�ϳɵĸ߷��ӻ�����M�Ļ�ѧ����ʽΪ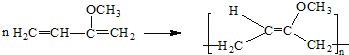
��ˣ�������ȷ������ ��
��
��6��A.��Ȳ��HCl���е����ʵ����ļӳɷ�Ӧ�õ�����ϩ��
B.����G�Ľṹ��ʽ����֪����G��������18����ԭ��������1mol G��ȫȼ������9mol H2O��
C.����F�Ľṹ��ʽ����֪����F����̼̼˫����������������F�ܷ����ӳɷ�Ӧ��ȡ����Ӧ��������Ӧ����ѡAC��

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�