��Ŀ����
 �����ô˸�������������ѧ���⣺
�����ô˸�������������ѧ���⣺��1����ͬ�����£���ʪ�������ܶ�________��ѡ�����������������������������ܶȡ�
��2����һ�������µĺ����ܱ������У���������ƽ�⣺5O2(g)��4NH3(g)
 4NO(g)��6H2O(g)�������¶ȱ��ֲ��䲢����He����ƽ��_______ ��������ƶ������������ƶ����������ƶ������� ����������� ______����д���������С�����䡱����
4NO(g)��6H2O(g)�������¶ȱ��ֲ��䲢����He����ƽ��_______ ��������ƶ������������ƶ����������ƶ������� ����������� ______����д���������С�����䡱������3����һ���¶��º����ܱ������У����������������ٱ仯ʱ�����ܱ�����Ӧ��A��s��+2B��g��
 C��g��+D��g���Ѵ�ƽ�����____________
C��g��+D��g���Ѵ�ƽ�����____________ A.��������ƽ����Է�������
B.���������ܶ�
C.B�����ʵ���Ũ��
D.����������ʵ���
��2�����ƶ�����С
��3��D

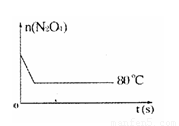
��80��ʱ��0.40mol��N2O4������� 2L�Ѿ���յĹ̶��ݻ����ܱ������У���һ��ʱ��Ը������ڵ����ʽ��з������õ��������ݣ�
| ������ʱ�䣨s�� n��mol�� | 0 | 20 | 40 | 60 | 80 | 100 |
| n��N2O4�� | 0.40 | a | 0.20 | c | d | e |
| n��NO2�� | 0.00 | 0.24 | b | 0.52 | 0.60 | 0.60 |
��1������20s��40s����N2O4��ʾ��ƽ����Ӧ����Ϊ������������������������������
��2�������� 80��ʱ�÷�Ӧ��ƽ�ⳣ��K���� ������������������������������
��3�����жϸ÷�Ӧ�Ƿ��Ѵﻯѧƽ��״̬��������________________��
a��������ѹǿ����
b�����������c(N2O4)����
c��v��(NO2)��v��(N2O4)
d����������ܶȲ���
e����������ƽ��Ħ����������
f��c(N2O4)= c(NO2)
��4����Ӧ������100s��Ӧ�������¶Ƚ��ͣ�����������ɫ�������������������������������dz������������䡱����
��5��Ҫ����÷�Ӧ��Kֵ���ɲ�ȡ�Ĵ�ʩ�У�����ţ�����������������Ҫ���´ﵽƽ��ʱ��ʹ[NO2]/[N2O4]ֵ��С���ɲ�ȡ�Ĵ�ʩ�У�����ţ�������������������������������
A������N2O4����ʼŨ�ȡ�������B������������ͨ��NO2
C��ʹ�ø�Ч�������������� D�������¶�
��6������ͼ�� 80��ʱ������N2O4���ʵ����ı仯���ߣ����ڸ�ͼ�в������÷�Ӧ�� 60��ʱN2O4���ʵ����ı仯���ߡ�
��80��ʱ��0.40mol��N2O4������� 2L�Ѿ���յĹ̶��ݻ����ܱ������У���һ��ʱ��Ը������ڵ����ʽ��з������õ��������ݣ�
|
������ʱ�䣨s�� n��mol�� |
0 |
20 |
40 |
60 |
80 |
100 |
|
n��N2O4�� |
0.40 |
a |
0.20 |
c |
d |
e |
|
n��NO2�� |
0.00 |
0.24 |
b |
0.52 |
0.60 |
0.60 |
��1������20s��40s����N2O4��ʾ��ƽ����Ӧ����Ϊ���� ��������������������������
��2�������� 80��ʱ�÷�Ӧ��ƽ�ⳣ��K���� ������������������������������
��3�����жϸ÷�Ӧ�Ƿ��Ѵﻯѧƽ��״̬��������________________��
a��������ѹǿ����
b�����������c(N2O4)����
c��v��(NO2)��v��(N2O4)
d����������ܶȲ���
e����������ƽ��Ħ����������
f��c(N2O4)= c(NO2)
��4����Ӧ������100s��Ӧ�������¶Ƚ��ͣ�����������ɫ�� �����������������������������dz������������䡱����
��5��Ҫ����÷�Ӧ��Kֵ���ɲ�ȡ�Ĵ�ʩ�У�����ţ�����������������Ҫ���´ﵽƽ��ʱ��ʹ[NO2]/[N2O4]ֵ��С���ɲ�ȡ�Ĵ�ʩ�У�����ţ�������������������������������
A������N2O4����ʼŨ�ȡ�������B������������ͨ��NO2
C��ʹ�ø�Ч�������������� D�������¶�
��6������ͼ�� 80��ʱ������N2O4���ʵ����ı仯���ߣ����ڸ�ͼ�в������÷�Ӧ�� 60��ʱN2O4���ʵ����ı仯���ߡ�

 H2 + CO2 + Q
H2 + CO2 + Q
 H2 + CO2 + Q
H2 + CO2 + Q