��Ŀ����
 �������ִ������а�����Խ��Խ��Ҫ�Ľ�ɫ������β����̼�⻯����������Pһ����̼�ȣ������Ļ�����ȾԽ��Խ���ԣ������������ŷ��ѳ�Ϊ���д�����Ⱦ����Ҫ��Դ��
�������ִ������а�����Խ��Խ��Ҫ�Ľ�ɫ������β����̼�⻯����������Pһ����̼�ȣ������Ļ�����ȾԽ��Խ���ԣ������������ŷ��ѳ�Ϊ���д�����Ⱦ����Ҫ��Դ����1�������ͳɷ�ΪC8H18�������������������ѽ��������Ͷ�ϩ��д���÷�Ӧ�Ļ�ѧ����
��2������������NO�ķ�ӦΪ��N2��g��+O2��g���T2NO��g����H��0
�������������������¶�Խ�ߣ���λʱ����NO�ŷ���Խ��ԭ����
��1mol�����к���0.8mol N2��0.2mol O2��1300��ʱ�ں�1mol�������ܱ������ڷ�Ӧ�ﵽƽ�⣮���NOΪ8��10-4mol��������¶��µ�ƽ�ⳣ��K��
��3��β���е�CO��Ҫ���������Ͳ���ȫȼ�գ�
���������밴���з�Ӧ��ȥCO��2CO��g���T2C��s��+O2��g����H=+221kJ?mol-1�������������ܷ�ʵ�ֵ����ݣ�
�ڲ�������β����Ũ�ȳ��õ绯ѧ����������������CO������������ͼ��ʾ�������������ĵ缫��ӦΪ
��4��������β��ϵͳ�а�װ��ת����������Ч����β���е�CO��NOx��̼�⻯����ȷ�����
��֪��N2��g��+O2��g���T2NO��g����H1=+180kJ/mol
CO��g��+
| 1 |
| 2 |
2NO��g��+2CO��g���T2CO2��g��+N2��g����H3
���H3=
��5���⻯ѧ��������������ȩ�ࡢ���������������ȣ�������β���������������·����⻯ѧ��Ӧ���ɵĶ�����Ⱦ�д��2-��ϩ����������ʵ���֮��Ϊ1��2��Ӧ������ȩ�������Ļ�ѧ����ʽ
���㣺��Ӧ�Ⱥ��ʱ�,�Ȼ�ѧ����ʽ,��ѧƽ��ļ���
ר�⣺�����������������
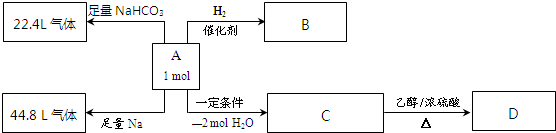
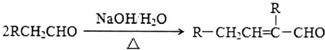
��������1���������ͷֽ����ɶ���Ͷ�ϩ�����ԭ���غ���д��ѧ���̣�
��2��������Ӱ�컯ѧ��Ӧ���ʺͻ�ѧƽ������ط����жϣ�
�ڽ�ϻ�ѧ����ʽ��ʽ����ƽ��Ũ�ȣ�ƽ�ⳣ������������ƽ��Ũ�ȵ��ݴη��˻����Է�Ӧ��ƽ��Ũ���ݴη��˻����㣻
��3�������ݷ�Ӧ�Է����е������ǡ�H-T��S��0�����жϣ�
�����ݵ��ԭ��������������ʧ���ӷ���������Ӧ�����ʣ����ͼʾ��֪��һ����̼ʧ�������ɶ�����̼��
��4�������Ȼ�ѧ����ʽ��˹���ɼ���õ������Ȼ�ѧ����ʽ���õ���Ӧ�ʱ䣻
��5��2-��ϩ����������ʵ���֮��Ϊ1��2��Ӧ������ȩ�����������ԭ���غ���д��ƽ��ѧ����ʽ��ע�ⷴӦ������
��2��������Ӱ�컯ѧ��Ӧ���ʺͻ�ѧƽ������ط����жϣ�
�ڽ�ϻ�ѧ����ʽ��ʽ����ƽ��Ũ�ȣ�ƽ�ⳣ������������ƽ��Ũ�ȵ��ݴη��˻����Է�Ӧ��ƽ��Ũ���ݴη��˻����㣻
��3�������ݷ�Ӧ�Է����е������ǡ�H-T��S��0�����жϣ�
�����ݵ��ԭ��������������ʧ���ӷ���������Ӧ�����ʣ����ͼʾ��֪��һ����̼ʧ�������ɶ�����̼��
��4�������Ȼ�ѧ����ʽ��˹���ɼ���õ������Ȼ�ѧ����ʽ���õ���Ӧ�ʱ䣻
��5��2-��ϩ����������ʵ���֮��Ϊ1��2��Ӧ������ȩ�����������ԭ���غ���д��ƽ��ѧ����ʽ��ע�ⷴӦ������
���
�⣺��1�������ͳɷ�ΪC8H18�������������������ѽ��������Ͷ�ϩ��д���÷�Ӧ�Ļ�ѧ����Ϊ��C8H18
C4H10+C4H8��
�ʴ�Ϊ��C8H18
C4H10+C4H8��
��2����N2��g��+O2��g���T2NO��g����H��0����Ӧʱ���������������ȷ�Ӧ���¶����ߣ���Ӧ���ʼӿ죬ƽ�����ƣ���λʱ����NO�ŷ���Խ��
�ʴ�Ϊ���¶����ߣ���Ӧ���ʼӿ죬ƽ�����ƣ�
��1mol�����к���0.8mol N2��0.2mol O2��1300��ʱ�ں�1mol�������ܱ������ڷ�Ӧ�ﵽƽ�⣮���NOΪ8��10-4mol��
N2��g��+O2��g���T2NO��g��
��ʼ����mol�� 0.8 0.2 0
�仯����mol�� 4��10-4 4��10-4 8��10-4
ƽ������mol�� 0.8-4��10-4 0.2-4��10-4 8��10-4
��Ӧǰ���������ʵ������䣬���������ʵ�������ƽ��Ũ�ȼ���ƽ�ⳣ��K=
��4��10-6��
�ʴ�Ϊ��4��10-6��
��3����2CO��g���T2C��s��+O2��g����H=+221kJ?mol-1����Ӧ���ʱ������ر��С�ķ�Ӧ����H��0����S��0�����H-T��S��0����Ӧ�κ��¶��¶������Է����У�
�ʴ�Ϊ���÷�Ӧ���������ؼ��ķ�Ӧ���κ��¶��¾������Է����У�
��������ʧ���ӷ���������Ӧ�����ʣ����ͼʾ��֪��һ����̼ʧ�������ɶ�����̼ͬʱ���������ӣ��缫��ӦΪ��CO+H2O-2e-=CO2+2H+��
�ʴ�Ϊ��CO+H2O-2e-=CO2+2H+��
��4����N2��g��+O2��g���T2NO��g����H1=+180kJ/mol
��CO��g��+
O2��g���TCO2��g����H2=-283kJ/mol
���ݸ�˹���ɼ���ڡ�2-�ٵõ���2NO��g��+2CO��g���T2CO2��g��+N2��g����H3 =-746KJ/mol��
�ʴ�Ϊ��-746��
��5��2-��ϩ����������ʵ���֮��Ϊ1��2��Ӧ������ȩ����������Ӧ�Ļ�ѧ����ʽΪ��CH3CH=CHCH3+2O3
2CH3CHO+2O2��
�ʴ�Ϊ��CH3CH=CHCH3+2O3
2CH3CHO+2O2��
| ���� |
�ʴ�Ϊ��C8H18
| ���� |
��2����N2��g��+O2��g���T2NO��g����H��0����Ӧʱ���������������ȷ�Ӧ���¶����ߣ���Ӧ���ʼӿ죬ƽ�����ƣ���λʱ����NO�ŷ���Խ��
�ʴ�Ϊ���¶����ߣ���Ӧ���ʼӿ죬ƽ�����ƣ�
��1mol�����к���0.8mol N2��0.2mol O2��1300��ʱ�ں�1mol�������ܱ������ڷ�Ӧ�ﵽƽ�⣮���NOΪ8��10-4mol��
N2��g��+O2��g���T2NO��g��
��ʼ����mol�� 0.8 0.2 0
�仯����mol�� 4��10-4 4��10-4 8��10-4
ƽ������mol�� 0.8-4��10-4 0.2-4��10-4 8��10-4
��Ӧǰ���������ʵ������䣬���������ʵ�������ƽ��Ũ�ȼ���ƽ�ⳣ��K=
| (8��10-4)2 |
| (0.8-4��10-4)(0.2-4��10-4) |
�ʴ�Ϊ��4��10-6��
��3����2CO��g���T2C��s��+O2��g����H=+221kJ?mol-1����Ӧ���ʱ������ر��С�ķ�Ӧ����H��0����S��0�����H-T��S��0����Ӧ�κ��¶��¶������Է����У�
�ʴ�Ϊ���÷�Ӧ���������ؼ��ķ�Ӧ���κ��¶��¾������Է����У�
��������ʧ���ӷ���������Ӧ�����ʣ����ͼʾ��֪��һ����̼ʧ�������ɶ�����̼ͬʱ���������ӣ��缫��ӦΪ��CO+H2O-2e-=CO2+2H+��
�ʴ�Ϊ��CO+H2O-2e-=CO2+2H+��
��4����N2��g��+O2��g���T2NO��g����H1=+180kJ/mol
��CO��g��+
| 1 |
| 2 |
���ݸ�˹���ɼ���ڡ�2-�ٵõ���2NO��g��+2CO��g���T2CO2��g��+N2��g����H3 =-746KJ/mol��
�ʴ�Ϊ��-746��
��5��2-��ϩ����������ʵ���֮��Ϊ1��2��Ӧ������ȩ����������Ӧ�Ļ�ѧ����ʽΪ��CH3CH=CHCH3+2O3
| ������ |
�ʴ�Ϊ��CH3CH=CHCH3+2O3
| ������ |
���������⿼���˻�ѧ����ʽ��д�����ԭ���ķ���Ӧ�ã���Ӧ�Է����е��ж����ݣ��Ȼ�ѧ����ʽ��˹���ɵļ���Ӧ�ã����ջ����ǹؼ�����Ŀ�ϼ�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
��NA��������ӵ���������ֵ������˵������ȷ���ǣ�������
| A�����³�ѹ�£�17g����-14CH3��������������Ϊ9NA |
| B��64g��ͭ����������ԭ��Ӧ��һ��ʧȥ2NA������ |
| C�����³�ѹ22.4L NO����ķ�����С��NA |
| D�����ʱ��22.4L���ȼ��������еķ�����ΪNA |
����˵����ȷ���ǣ�������
| A�����ʯī������Ҫ�İ뵼����� |
| B��SO2��NO2��CO2���ᵼ��������γ� |
| C��������Ư�۳���������ˮ�ľ�����ɱ�����������ߵ�����ԭ����ͬ |
| D�������������Դ����ҵ���õ��ˮ���ƴ����������Ͻ��ܼ������� |
��������������Һ�����ܸ�пƬ��Ӧ�����������Ӧ���������ǣ�������
| A��10��10mL 4mol/L��������Һ |
| B��20��10mL 4mol/L��������Һ |
| C��20��20mL 2mol/L��������Һ |
| D��20��40mL 2mol/L��������Һ |
����ͼʾ���Ӧ������������ǣ�������


| A��ͼ���ʾ����μӵ�0.1mol/Lij����Һ�еõ��ĵζ����ߣ���ͼ���֪����ǡ���к�ʱ��������Һ��pH��7 |
| B��ͼ���ʾһ�������½��еķ�Ӧ2SO2+O2?2SO3��H��0���ɷֵ����ʵ����仯��t2ʱ�̸ı�����������������¶� |
| C��ͼ���ʾij������Һ�м���Ba��OH��2��Һ�����������������Ba��OH��2��Һ����Ĺ�ϵ���ڼ���20mL Ba��OH��2��Һʱ������ȫ����BaSO4 |
| D��ͼ����ʾpH��ͬ������������зֱ����ˮ����ҺpH�ı仯����������a��Ӧ�������� |
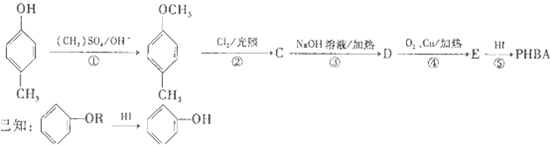
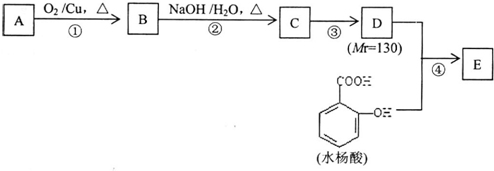
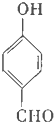 �Ի�����ȩ�����׳�PHBA����-����Ҫ���л�����ԭ�ϣ���ṹ��ͼ��ʾ
�Ի�����ȩ�����׳�PHBA����-����Ҫ���л�����ԭ�ϣ���ṹ��ͼ��ʾ