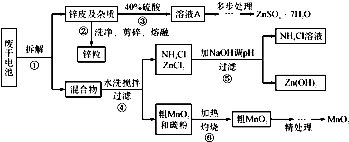
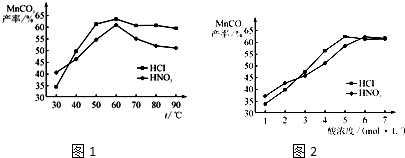
��Ŀ����
��֪25��ʱij��Һ�к��д���Na+��H+��Fe3+��HC03-��OH-��I-�еļ��֣�����ˮ �������c��H+��=1��lO-13 mol/L���������Һ�л���ͨ��һ������Cl2����Һ����ɫ ��Ϊ��ɫ�������йط�����ȷ���ǣ�������
| A����Һ��pH=1��13 | B����Һ��һ��û��Fe3+��Na+ | C����Һ����������I-������ȷ��HCO3- | D����Cl2������������Һֻ���������� |
������ˮ�������c��H+��=1��lO-13 mol/L����Һ�������ӻ�����������Ũ��Ϊ0.1mol/L������ҺΪǿ���Ի������Һ��һ���������̼��������ӣ��������Һ�л���ͨ��һ������Cl2����Һ����ɫ��Ϊ��ɫ��˵����Һ��һ�����ڵ����ӣ��ܹ�������ӷ�Ӧ��������һ��������ڣ��ݴ���ɱ��⣮
����⣺��ˮ�������c��H+��=1��lO-13 mol/L������Һ�д������������ӻ������������ӣ�������Һ��һ���������̼��������ӣ�ͨ����������Һ����ɫ ��Ϊ��ɫ��˵����Һ��һ�����������ɫ�������ӣ�һ�����ڵ����ӣ�
A�����ݷ�����֪��ˮ�������c��H+��=1��lO-13 mol/L����Ϊ������Һ������ҺpH=1����Ϊ������Һ������Һ��pHΪ13����A��ȷ��
B����Һ��һ��������������ӣ����Ǽ�����Һ��һ�����������ӣ���B����
C������Һ��һ�����ڵ����ӣ�һ���������̼��������ӣ���C����
D��������������Ӧ��ֻ��һ���Σ����Ȼ��ƣ���D����
��ѡA��
A�����ݷ�����֪��ˮ�������c��H+��=1��lO-13 mol/L����Ϊ������Һ������ҺpH=1����Ϊ������Һ������Һ��pHΪ13����A��ȷ��
B����Һ��һ��������������ӣ����Ǽ�����Һ��һ�����������ӣ���B����
C������Һ��һ�����ڵ����ӣ�һ���������̼��������ӣ���C����
D��������������Ӧ��ֻ��һ���Σ����Ȼ��ƣ���D����
��ѡA��
���������⿼���˳������ӵļ��鷽������Ŀ�Ѷ��еȣ�ע����ȷ�������ӵķ�Ӧ�����鷽������������������ѧ���ķ������������������Ӧ����ѧ֪ʶ��������

��ϰ��ϵ�д�
 ������ʱ����ҵ����ϵ�д�
������ʱ����ҵ����ϵ�д� ��ĩ���ƾ�ϵ�д�
��ĩ���ƾ�ϵ�д� ���ɿ��ñ���ϵ�д�
���ɿ��ñ���ϵ�д�
�����Ŀ
 ��2013?������ģ����ҵ�ϡ��̶���������CO2����Ч�ؼ��ᡰ����ЧӦ����
��2013?������ģ����ҵ�ϡ��̶���������CO2����Ч�ؼ��ᡰ����ЧӦ����