��Ŀ����
����Ŀ������23V)���ҹ��ķ��Ԫ�أ��㷺���ڴ���������ҵ���ش��������⣺
��1������Ԫ�����ڱ��е�λ��Ϊ_______����۲�����Ų�ͼΪ____________��
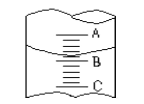
��2������ij��������ľ����ṹ��ͼ1��ʾ��������ʵ��ӵ�е����������Ӹ����ֱ�Ϊ____��_____��
��3��V2O5������SO2ת��ΪSO3�Ĵ�����SO2������Sԭ�Ӽ۲���Ӷ�����__�ԣ����ӵ����幹��Ϊ___��SO3��̬Ϊ�����ӣ��÷�����Sԭ�ӵ��ӻ��������Ϊ____��SO3�������廷״�ṹ��ͼ2��ʾ���ýṹ��Sԭ�ӵ��ӻ��������Ϊ___���ýṹ��S��O���������࣬һ�����Լ140pm����һ�����ԼΪ160pm���϶̵ļ�Ϊ__����ͼ2����ĸ)���÷����к���___��������
��4��V2O5�ܽ���NaOH��Һ�У��ɵõ������ƣ�Na3VO4)�����������ӵ����幹��Ϊ___��Ҳ���Եõ�ƫ�����ƣ��������ӳ���ͼ3��ʾ��������״�ṹ����ƫ�����ƵĻ�ѧʽΪ_______��
���𰸡� ��4������B�� ![]() 4 2 3 V�� sp2�ӻ� sp3�ӻ� a 12 ���������� NaVO3
4 2 3 V�� sp2�ӻ� sp3�ӻ� a 12 ���������� NaVO3
�������������������1��������֪�����ĺ˵����Ϊ23���������֪����Ԫ�����ڱ��е�λ��Ϊ��4������A�壬���ݺ�����ӵĹ�������Ų�˳�����ƶ�������Ų�ʽΪ1s22s22p63s23p63d34s2������d���������ڵ������ڵ���B�壬��۲�����Ų�ͼΪ![]() ��
��
��2���ɾ�����֪��Vλ�ڶ�������ģ������Ӹ���Ϊ1+8��1/8=2��O��4��λ�����ģ�2��λ�����ģ��������Ӹ���Ϊ4��1/2+2=4�����Ծ�����ʵ��ӵ�е����������Ӹ����ֱ�Ϊ4,2.
��3��SO2������Sԭ���γ�2�������µ��Ӷ���Ϊ��6-2��2����2=1��SO2������Sԭ�Ӽ۲���Ӷ�����3��ΪV�νṹ��SO3��̬Ϊ�����ӣ��÷�����Sԭ���γ�3������û�й¶Ե��ӣ���Ϊsp2�ӻ���SO3����������Sԭ���γ�4������Ϊsp3�ӻ���SO3����������ÿ��S����S=O����S-O����S=O�����϶̣���a�϶̣��÷����к�������ĿΪ3��4=12
��4��VO43-�У�V�γ�4�������µ��Ӷ���Ϊ��5+3-4��2����2=0��Ϊ��������ṹ������״�ṹ��֪ÿ��V��3��O�γ������ӣ���V�Ļ��ϼ�Ϊ+5�ۣ����γɵĻ����ﻯѧʽΪNaVO3��

 ����ѧУ�ֲ����ܲ�ϵ�д�
����ѧУ�ֲ����ܲ�ϵ�д� �ƸԺ���ȫ�����Ų��Ծ�ϵ�д�
�ƸԺ���ȫ�����Ų��Ծ�ϵ�д�����Ŀ������������Ҫ������NO2��SO2��ij����С����������о���
��1��������������ͨ��������ˮ�а������������ᣬ����NH3��H2O���ա�
��֪2NH3��H2O (aq) + H2SO4(aq)==(NH4)2SO4(aq)+ 2H2O(l) ��H=-24.2kJ/mol���к�����H=-57.3 kJ/mol��1mol NH3��H2O��������ȫ����ʱ��Ҫ���յ�����Ϊ________��
��2��������Ҳ������NaOH��Һֱ������SO2�������չ����У���ҺpH��n(SO32-):n(HSO3-)�仯��ϵ���±�:
n(SO32-):n(HSO3-) | 88:12 | 1:1 | 12:88 |
pH | 8.2 | 7.4 | 6.0 |
��������Һ������ʱ����Һ������Ũ���ɴ�С����˳��Ϊ___________��
������NaOH��Һ��ͨ��������SO2ʱ���õ�NaHSO3��Һ����pHΪ4~7֮���⣬��Ԫ���������ϱ���ԭΪNa2S2O4�����ǵ绯ѧ������֮һ��д���������ĵ缫��Ӧʽ_______��
��3�����ݻ�Ϊ2L���ܱ������зֱ����0.20molNO2��0.4mol SO2������SO2(g)+NO2(g) ![]() SO3(g)+NO(g) ��H=-41.8 kJ/mol �ڲ�ͬ�¶��²ⶨͬһʱ��NO2��ת���ʣ��������ͼ��ʾ��
SO3(g)+NO(g) ��H=-41.8 kJ/mol �ڲ�ͬ�¶��²ⶨͬһʱ��NO2��ת���ʣ��������ͼ��ʾ��

��a��c���㷴Ӧ���ʴ�С��ϵ: v(a) ___v(c)��(����>������<�� ����=��)
���¶�ΪT2ʱ�ӷ�Ӧ��ʼ����2min �ﵽb�㣬��SO3��ʾ���ʱ��ķ�Ӧ����Ϊ______�����¶��¸÷�Ӧ��ƽ�ⳣ��Ϊ_______�����ڴ��¶��£������������ݻ����䣬���������г���0.10molNO2��0.2molSO2��NO2��ת����_______(����������������С������������).
��NO2��ת�������¶�������������С��ԭ����____________��