��Ŀ����
��֪��A��B��C��D��E��F����Ԫ�أ�ԭ��������������Aԭ�Ӻ�����������״�ĵ����ƣ�������״�ĵ����ƹ���ϵ�������ȣ�B�Ƕ�������ԭ�Ӱ뾶����Ԫ�أ�CԪ��3p�ܼ��������E���������ڵ縺������Ԫ�أ�F�ǵ�������δ�ɶԵ�������Ԫ�ء��Իش������йص����⣺
��1��д��FԪ�صĵ����Ų�ʽ_______��
��2����֪AԪ�ص�һ���⻯������к��ĸ�ԭ�ӣ����ڸû�����ķ�����Aԭ�ӵ��ӻ��������Ϊ______��
��3����֪C��E����Ԫ���γɵĻ�����ͨ����CE3��CE5���֡������ֻ�������һ��Ϊ�Ǽ��Է��ӣ�һ��Ϊ���Է��ӣ����ڼ��Է��ӵĻ�����ķ��ӿռ乹����______��
��4��B��C��D��E�ĵ�һ�������ɴ�С��˳����______ (дԪ�ط��ţ�������Ԫ������������ˮ�����γɵ���Һ�����ʵ���Ũ����ͬʱ��pH�ɴ�С��˳����______(д��ѧʽ����
��5����B��E��Ԫ���γɵĻ�������ɵľ����У����������Ӷ������ͶԳƽṹ�����Ƕ����Կ�������Բ���˴ˡ����С�������ͼ��ʾΪB��E�γɻ�����ľ����ṹͼ�Լ�����������ͼ��

�����о���һ��Na+�����Na+��____________�����������ܶ�Ϊ�� g��cm-3�������ӵ�����ֵ��NA��ʾ����Cl�������Ӱ뾶_____cm(�ú�NA���ʽ�ӱ���)��
��1��[Ar]3d54s1
��2��sp3
��3��������
��4��Cl�� P�� S��Na NaOH �� H3PO4 ��HClO4��H2SO4
��5��12 
��������
���������Aԭ�Ӻ�����������״�ĵ����ƣ�������״�ĵ����ƹ���ϵ�������ȣ����̬Aԭ�ӵĺ�������Ų�Ϊ1s22s22p4��AΪO��B�Ƕ�������ԭ�Ӱ뾶����Ԫ�أ���BΪNa��CԪ��3p�ܼ�����������̬Cԭ�ӵĺ�������Ų�Ϊ1s22s22p63s23p3��CΪP��F�ڵ������ڣ���F��ԭ����������E��E�����������ڵ縺������Ԫ�أ���E�ڵ������ڣ�EΪCl��D��ԭ����������C��С��E����DΪS��F�ǵ�������δ�ɶԵ�������Ԫ�أ����̬Fԭ�ӵĺ�������Ų�Ϊ1s22s22p63s23p63d54s1����FΪCr��
��1����̬Fԭ�ӵĺ�������Ų�Ϊ1s22s22p63s23p63d54s1����[Ar]3d54s1��
��2��A��һ���⻯�ﺬ��4��ԭ�ӣ����⻯��ΪH2O2�� O����㹲��4�Ե��ӣ��ڿռ���4����չ���ʹ���ӻ�����Ϊsp3��
��3��P��Cl�γ�PCl3��PCl5���ֻ��������PCl3�Ǽ��Է��ӣ�����ԭ��P�������5�����ӣ���3���ֱ���3��Cl�γɹ��õ��Ӷԣ���ʣ��һ���µ��Ӷԣ����P���ӻ���ʽΪsp3����PCl3�ķ��ӹ�����NH3���ƣ��������͡�
��4��ͬһ����Ԫ�أ���һ������������������������ƣ�����P��3p������ڰ����״̬���Ƚ��ȶ�����˵�һ������P��S����ͬŨ�ȵ�������Һ�������Ƕ�Ԫǿ�ᣬ��Һ��c(H+)���pH��С��H3PO4����ǿ�ᣬ������ȫ�����H+�����Ա�HClO4�������pH����HClO4��
��5����NaCl�����У�����һ��Na+�����Na+��12�����ֱ���x��y��z��������ƽ����ĸ����㡣
ÿ��NaCl�����к���4��NaCl���辧�����ⳤΪa cm������ ��a=
��a=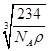 ��Cl-�İ뾶����Na+�İ뾶�����ݾ�������ͼ����֪��������ĶԽ��߳�ΪCl-�뾶��4������Խ��߳�Ϊ
��Cl-�İ뾶����Na+�İ뾶�����ݾ�������ͼ����֪��������ĶԽ��߳�ΪCl-�뾶��4������Խ��߳�Ϊ ����Cl-�İ뾶Ϊ
����Cl-�İ뾶Ϊ ��
��
���㣺����ԭ�Ӻ�������Ų�����һ�����ܣ�ԭ�ӹ���ӻ������ӹ��ͣ������ļ���ȡ�

 ��ɢ˼ά�¿���ϵ�д�
��ɢ˼ά�¿���ϵ�д� ��2011?�����ģ����ѧ--ѡ�����ʽṹ������
��2011?�����ģ����ѧ--ѡ�����ʽṹ������
 ��֪��A��B��C��D��E��F��XΪ���ڱ���ǰ�����ڵ�����Ԫ�أ����ǵ�ԭ��������������A�����ڱ���ԭ�Ӱ뾶��С��Ԫ�أ�B�Ļ�̬ԭ����3����ͬ���ܼ������ܼ��е�������ȣ�D�Ļ�̬ԭ��2p�ܼ��ϵ�δ�ɶԵ�������Bԭ�ӵ���ͬ��D2-������E2+���Ӿ�����ͬ���ȶ����Ӳ�ṹ��F�С����������֮�ƣ�F4+���Ӻ��ԭ�ӵĺ�������Ų���ͬ��X�Ļ�̬ԭ�ӵļ۵����Ų�ʽΪ3d84s2��
��֪��A��B��C��D��E��F��XΪ���ڱ���ǰ�����ڵ�����Ԫ�أ����ǵ�ԭ��������������A�����ڱ���ԭ�Ӱ뾶��С��Ԫ�أ�B�Ļ�̬ԭ����3����ͬ���ܼ������ܼ��е�������ȣ�D�Ļ�̬ԭ��2p�ܼ��ϵ�δ�ɶԵ�������Bԭ�ӵ���ͬ��D2-������E2+���Ӿ�����ͬ���ȶ����Ӳ�ṹ��F�С����������֮�ƣ�F4+���Ӻ��ԭ�ӵĺ�������Ų���ͬ��X�Ļ�̬ԭ�ӵļ۵����Ų�ʽΪ3d84s2��
 ��֪����A��B��C��D�����µķ�Ӧ��ϵ������A ��B��ȼ��ʱ������ʲ�ɫ�� C��B��ȼ��ʱ�����ػ�ɫ���̣�E��ˮ��Һ������ɫ��G��һ�ֺ�ɫ���壮
��֪����A��B��C��D�����µķ�Ӧ��ϵ������A ��B��ȼ��ʱ������ʲ�ɫ�� C��B��ȼ��ʱ�����ػ�ɫ���̣�E��ˮ��Һ������ɫ��G��һ�ֺ�ɫ���壮