��Ŀ����
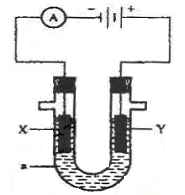
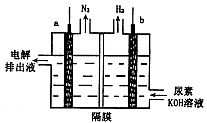
��ҵ�������������õ����е�ⱥ��ʳ��ˮ��Ϊ�˱��������֮�䷢����Ӧ�����������ӽ���Ĥ�����۸��������֡���ͼ����ͼ��Ϊ���۵�ʾ��ͼ��
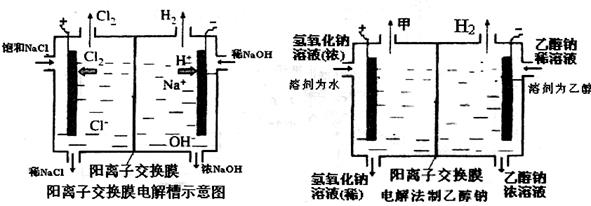
��1�����������ӽ���Ĥ��ֻ������Һ�е� ͨ���������������ı�ţ�
��H2 ��Cl2 ��H+ ��Cl�� ��Na+ ��OH��
��2��д���ڵ������з����ĵ缫����ʽ������ ��
��3����֪ij����ÿСʱ����10%������������Һ10kg��ÿСʱ���ռ�������µ�����896L���������ߵ�ˮ����������ͨ���������ϼ��㣬��������������������Һ����������Ϊ ��
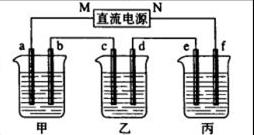
��4��ij��ѧ������ȤС��������õ�ⷨ��ȡ�Ҵ��ƵĹ�ҵ���������õĵ���������ͼ��ʾ�����Ҫ�����õĽ���Ĥ�����÷�������ͨ�����ڵ����������ĵ�ԭ�����������ƺ��Ҵ����ش��������⣺
��д���ڵ������з����ĵ缫����ʽ
������ ��
�������Ҵ��Ƶ��Ҵ���Һ�з���õ������Ҵ��ƹ���ķ����ǣ� ��

��1�����������ӽ���Ĥ��ֻ������Һ�е� ͨ���������������ı�ţ�
��H2 ��Cl2 ��H+ ��Cl�� ��Na+ ��OH��
��2��д���ڵ������з����ĵ缫����ʽ������ ��
��3����֪ij����ÿСʱ����10%������������Һ10kg��ÿСʱ���ռ�������µ�����896L���������ߵ�ˮ����������ͨ���������ϼ��㣬��������������������Һ����������Ϊ ��
��4��ij��ѧ������ȤС��������õ�ⷨ��ȡ�Ҵ��ƵĹ�ҵ���������õĵ���������ͼ��ʾ�����Ҫ�����õĽ���Ĥ�����÷�������ͨ�����ڵ����������ĵ�ԭ�����������ƺ��Ҵ����ش��������⣺
��д���ڵ������з����ĵ缫����ʽ
������ ��
�������Ҵ��Ƶ��Ҵ���Һ�з���õ������Ҵ��ƹ���ķ����ǣ� ��
��1���ۢݣ�3�֣���2��������2Cl����2e��=Cl2��3�֣� ��3��35.7%��3�֣�
��4����������2CH3CH2OH+2e��=2CH3CH2O��+H2��3�֣��������ᾧ ��3�֣�
��4����������2CH3CH2OH+2e��=2CH3CH2O��+H2��3�֣��������ᾧ ��3�֣�
�����������1�������ӽ���Ĥֻ���������������ӡ�������ͨ�����������Ӻͷ��Ӿ�����ͨ�����ʴ�Ϊ���ۢݡ�
��2�������к͵�Դ�������������ǵ��ص������������������ӷ���ʧ���ӵ�������Ӧ����2Cl--2e-=Cl2�����ʴ�Ϊ��2Cl--2e-=Cl2����
��3�����������ʵ���=896L��22.4L/mol=40mol������Ϊ40mol��2g/mol=80g�����ݵ�ⷽ��ʽ��2NaCl+2H2O
 2NaOH+H2��+Cl2�����ɵ����ɵ�NaOHΪ��m(NaOH)=2��40mol��40g/mol=3200g����Һ���ӵ�����Ϊ���ӵ���Ԫ�ص�������ȥ����������������Ϊ��2��40mol��23g/mol-80g=1760g����Һ�������������Ƶ�����Ϊ10000g��10%+3200g=4200g����Һ������Ϊ10000g+1760g=11760g����������������������Һ����������=4200g��11760g��100%=35.7%��
2NaOH+H2��+Cl2�����ɵ����ɵ�NaOHΪ��m(NaOH)=2��40mol��40g/mol=3200g����Һ���ӵ�����Ϊ���ӵ���Ԫ�ص�������ȥ����������������Ϊ��2��40mol��23g/mol-80g=1760g����Һ�������������Ƶ�����Ϊ10000g��10%+3200g=4200g����Һ������Ϊ10000g+1760g=11760g����������������������Һ����������=4200g��11760g��100%=35.7%����4���ٵ����к͵�Դ�ĸ����������ǵ��ص��������������Ҵ��õ��ӷ�����ԭ��Ӧ����2CH3CH2OH+2e-=2CH3CH2O-+H2����
�ڴ���Һ���������ʵķ����������ᾧ���ʴ�Ϊ�������ᾧ��

��ϰ��ϵ�д�
�����Ŀ

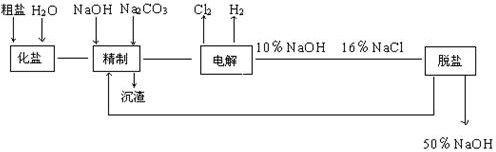
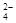 �����ϸߣ��������ӱ�ʽ����ȥSO
�����ϸߣ��������ӱ�ʽ����ȥSO