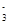��Ŀ����
ʵ����Ϊ�������й������ĺ�������������Ϳ��CuI����ֽ��������ֽ�Ƿ��ɫ����ɫ�����仯����ȥ��ʱ�����жϿ����еĺ��������䷴ӦΪ��4CuI��Hg��Cu2HgI4��2Cu
��1��������Ӧ����Cu2HgI4�У�CuԪ���� �ۡ�
��2�����Ϸ�Ӧ�е�������Ϊ ������1 mol CuI���뷴Ӧʱ��ת�Ƶ��� mol��
��3��CuI����Cu2����I��ֱ�ӷ�Ӧ�Ƶã�����ƽ���з�Ӧ�����ӷ���ʽ��
Cu2�� �� I���� CuI�� I3��
��1����1 ��2��CuI 0.5 ��3��2 5 2 1

��ϰ��ϵ�д�
 ���ɿ��õ�Ԫ����AB��ϵ�д�
���ɿ��õ�Ԫ����AB��ϵ�д� С�����ϵ�д�
С�����ϵ�д�
�����Ŀ
ʵ����Ϊ�������й������ĺ�������������Ϳ��CuI����ֽ��������ֽ����ɫ�����仯����ȥ��ʱ�����жϿ����еĺ��������䷴ӦΪ��4CuI+Hg�TCu2HgI4+2Cu������˵����ȷ���ǣ�������
| A��CuI�ڷ�Ӧ��ʧȥ���� | B��Cu2HgI4��CuԪ�صĻ��ϼ�Ϊ+2 | C��CuI�������� | D��1mol CuI���뷴Ӧ��1mol����ת�� |
Һ̬�����������¼����������������о綾��ʵ����Ϊ�������й������ĺ�����ͨ��������CuI����ֽ��������ֽ�Ƿ��ɫ����ɫ�����仯��ʱ�����жϿ����й��ĺ������䷴ӦΪ��4CuI+Hg�TCu2HgI4+2Cu��I�Ļ��ϼ۷�Ӧǰ���Ϊ-1�۵ģ��������й�˵������ȷ���ǣ�������
| A��������Ӧ�IJ���Cu2HgI4�У�Hg�Ļ��ϼ�Ϊ+2 | B��������Ӧ����������CuI | C��������Ӧ��CuԪ�ؽ���һ�뱻��ԭ | D��������Ӧ������64g Cuʱ��ת�Ƶĵ�����Ϊ2NA |
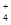 ��Cl����OH����CO
��Cl����OH����CO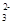 B��Fe2����NO3-��Cl����H��
B��Fe2����NO3-��Cl����H��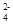
 Cu2����
Cu2����