��Ŀ����
| ʵ�� ��� |
HA���ʵ��� Ũ��/��mol?L-1�� |
NaOH���ʵ��� Ũ��/��mol?L-1�� |
��Ϻ��� Һ��pH |
| �� | 0.1 | 0.1 | pH=a |
| �� | 0.12 | 0.1 | pH=7 |
| �� | 0.2 | 0.1 | pH��7 |
| �� | 0.1 | 0.1 | pH=10 |
��2����������Һ������Ũ��c��A-����c��Na+���Ĵ�С��ϵ
A��ǰ�ߴ� B�����ߴ�
C��������� D�����ж�
��3���ӱ���ʵ�����������û����Һ������Ũ���ɴ�С��˳����
��4����������ʵ�����ݣ�д�������Һ��������ʽ�ľ�ȷ�������ʽ����
c��Na+��-c��A-��=
����ij��Ԫ�ᣨ��ѧʽ��H2B��ʾ����ˮ�еĵ��뷽��ʽ�ǣ�H2B�TH++HB-��HB-?H++B2-
�ش��������⣺
��5����0.1mol?L-1��Na2B��Һ�У���������Ũ�ȹ�ϵʽ��ȷ����
A��c��B2-��+c��HB-��=0.1mol?L-1
B��c��B2-��+c��HB-��+c��H2B��=0.1mol?L-1
C��c��OH-��=c��H+��+c��HB-��
D��c��Na+��+c��H+��=c��OH-��+c��HB-��
��2���κ���Һ�ж����ڵ���غ㣬���ݵ���غ��жϣ�
��3�������Һ������Ϊ�����ʵ�����HA��NaA��pH��7˵��A-��ˮ�����HA�ĵ��룬��ϵ���غ��жϣ�
��4���ɵ���غ��ϵʽ���ε�c��Na+��-c��A-��=c��OH-��-c��H+����
��II�����ݶ�Ԫ��ĵ��뷽��ʽ֪��B2-ֻ������һ��ˮ�⣬��ϵ���غ�������غ�������
��2�������Һ�д��ڵ���غ�c��Na+��+c��H+��=c��A-��+c��OH-��������pH=7����c��Na+��=c��A-����
�ʴ�Ϊ��C��
��3�������Һ������Ϊ�����ʵ�����HA��NaA��pH��7˵��A-��ˮ�����HA�ĵ��룬��������Ũ���ɴ�С��˳��Ϊc��Na+����c��A-����c��OH-����c��H+����
��4���ɵ���غ��ϵʽ���ε�c��Na+��-c��A-��=c��OH-��-c��H+��=��10-4-10-10��mol?L-1��
�ʴ�Ϊ��10-4-10-10��
��5����Na2B�д���ˮ��ƽ�⣺B2-+H2O=HB-+OH-��HB-�����һ��ˮ�⣬������Һ��û��H2B���ӣ�
A�����������غ��c��B2-��+c��HB-��=0.1mol?L-1����A��ȷ��
B��HB-�����һ��ˮ�⣬������Һ��û��H2B���ӣ���B����
C�����������غ��c��OH-��=c��H+��+c��HB-������C��ȷ��
D�����ݵ���غ��c��Na+��+c��H+��=c��OH-��+c��HB-��+2c��B2-������D����
�ʴ�Ϊ��AC��

 �������ϵ�д�
�������ϵ�д���2013?����һģ�������£���ijһԪ��HA��NaOH��Һ�������ϣ�ʵ����Ϣ���£�
|
�����£���ijһԪ��HA��NaOH��Һ�������ϣ�������Һ�����ʵ���Ũ�Ⱥͻ�Ϻ�������Һ��pH���±���
| ʵ���� | HA��Ũ��/��mol/L�� | NaOH��Ũ��/��mol/L�� | �����Һ��pH |
| �� | 0.2 | 0.2 | pH=a |
| �� | c | 0.2 | pH=7 |
| �� | 0.2 | 0.1 | pH��7 |
| �� | 0.1 | 0.1 | pH=9 |
��ش�
��1��������������ʵ���������Ӽ��������������a 7(�����������������)��HAΪ���
��2���������������ʵ���������������������������������һ���������
A.����Һ�����ʵ���Ũ�ȴ��ڼ���Һ
B.����Һ��H+��Ũ�ȴ��ڼ���Һ��OH����Ũ��
C.����Һ�����ʵ���Ũ��С�ڼ���Һ
D.����Һ��H+��Ũ��С�ڼ���Һ��OH����Ũ��
E.������Һ�����ʵ���Ũ�����
��3���ӱ���ʵ����������HA�� �ᣨ�ǿ������������ ����ʱ10mL0.5mol/LNaA��Һ��6mL1mol/L�����Ϻ���Һ�г�OH����ĸ�����Ũ���ɴ�С˳��Ϊ ��
��4������ʵ�����û����Һ����ˮ���������c(OH��)= mol/L��
��5������HA��һԪ���ᣬ��������CaA2��ˮ�д����ܽ�ƽ�⣺CaA2(s) ![]() Ca2++2A-
Ca2++2A-
��H��0��һ���¶���CaA2������Һ��Ksp=c(Ca2+)c2(A2-)Ϊһ������
���¶�����ʱ��Ksp �����������С�����䡱����
�ڲ��25��ʱ��CaA2��KspΪ4.0��10-11�������½�10g CaA2����Ͷ��100mLCaCl2��Һ�У���ֽ�������й���ʣ�࣬�����Һ��c(Ca2+)=0.1mol/L������Һ��c(A2-)= ��
��ƽ�ⳣ�������˷����ϵ�Ŀ��淴Ӧ�ڸ������¶��½��еij̶ȣ�����ͬһ�����͵ķ�Ӧ��ƽ�ⳣ��Խ������Ӧ���еij̶�Խ��
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
����������̼��ʹ�����ĵ���ƽ�ⳣ����д����������������������Ӧ�����ӷ���ʽ��
(1)������������ͨ�뵽������̼������Һ��___ _____________��
(2)������̼���ư���1�U1�����ʵ���֮��ǡ�÷�Ӧ_______________________________��
�����£���ijһԪ��HA��NaOH��Һ�������ϣ�������Һ�����ʵ���Ũ�Ⱥͻ�Ϻ�������Һ��pH���±���
| ʵ���� | HA��Ũ��/��mol/L�� | NaOH��Ũ��/��mol/L�� | �����Һ��pH |
| �� | 0.2 | 0.2 | pH=a |
| �� | c | 0.2 | pH=7 |
| �� | 0.2 | 0.1 | pH��7 |
| �� | 0.1 | 0.1 | pH=9 |
��ش�
��1��������������ʵ���������Ӽ��������������a 7(�����������������)��HAΪ���
��2���������������ʵ���������������������������������һ���������
A.����Һ�����ʵ���Ũ�ȴ��ڼ���Һ
B.����Һ��H+��Ũ�ȴ��ڼ���Һ��OH����Ũ��
C.����Һ�����ʵ���Ũ��С�ڼ���Һ
D.����Һ��H+��Ũ��С�ڼ���Һ��OH����Ũ��
E.������Һ�����ʵ���Ũ�����
��3���ӱ���ʵ����������HA�� �ᣨ�ǿ������������ ����ʱ10mL0.5mol/LNaA��Һ��6mL1mol/L�����Ϻ���Һ�г�OH����ĸ�����Ũ���ɴ�С˳��Ϊ ��
��4������ʵ�����û����Һ����ˮ���������c(OH��)= mol/L��
��5������HA��һԪ���ᣬ��������CaA2��ˮ�д����ܽ�ƽ�⣺CaA2(s) ![]() Ca2++2A-
Ca2++2A-
��H��0��һ���¶���CaA2������Һ��Ksp=c(Ca2+)c2(A2-)Ϊһ������
���¶�����ʱ��Ksp �����������С�����䡱����
�ڲ��25��ʱ��CaA2��KspΪ4.0��10-11�������½�10g CaA2����Ͷ��100mLCaCl2��Һ�У���ֽ�������й���ʣ�࣬�����Һ��c(Ca2+)=0.1mol/L������Һ��c(A2-)= ��
��ƽ�ⳣ�������˷����ϵ�Ŀ��淴Ӧ�ڸ������¶��½��еij̶ȣ�����ͬһ�����͵ķ�Ӧ��ƽ�ⳣ��Խ������Ӧ���еij̶�Խ��
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
����������̼��ʹ�����ĵ���ƽ�ⳣ����д����������������������Ӧ�����ӷ���ʽ��
(1)������������ͨ�뵽������̼������Һ��___ _____________��
(2)������̼���ư���1�U1�����ʵ���֮��ǡ�÷�Ӧ_______________________________��
�����£���ijһԪ��HA��NaOH��Һ�������ϣ�������Һ�����ʵ���Ũ�Ⱥͻ�Ϻ�������Һ��pH���±���
|
ʵ���� |
HA��Ũ��/��mol/L�� |
NaOH��Ũ��/��mol/L�� |
�����Һ��pH |
|
�� |
0.2 |
0.2 |
pH=a |
|
�� |
c |
0.2 |
pH=7 |
|
�� |
0.2 |
0.1 |
pH��7 |
|
�� |
0.1 |
0.1 |
pH=9 |
��ش�
��1��������������ʵ���������Ӽ��������������a 7(�����������������)��HAΪ���
��2���������������ʵ���������������������������������һ���������
A.����Һ�����ʵ���Ũ�ȴ��ڼ���Һ
B.����Һ��H+��Ũ�ȴ��ڼ���Һ��OH����Ũ��
C.����Һ�����ʵ���Ũ��С�ڼ���Һ
D.����Һ��H+��Ũ��С�ڼ���Һ��OH����Ũ��
E.������Һ�����ʵ���Ũ�����
��3���ӱ���ʵ����������HA�� �ᣨ�ǿ������������ ����ʱ10mL0.5mol/LNaA��Һ��6mL1mol/L�����Ϻ���Һ�г�OH����ĸ�����Ũ���ɴ�С˳��Ϊ ��
��4������ʵ�����û����Һ����ˮ���������c(OH��)= mol/L��
��5������HA��һԪ���ᣬ��������CaA2��ˮ�д����ܽ�ƽ�⣺CaA2(s)  Ca2++2A-
Ca2++2A-
��H��0��һ���¶���CaA2������Һ��Ksp=c(Ca2+)c2(A2-)Ϊһ������
���¶�����ʱ��Ksp �����������С�����䡱����
�ڲ��25��ʱ��CaA2��KspΪ4.0��10-11�������½�10g CaA2����Ͷ��100mLCaCl2��Һ�У���ֽ�������й���ʣ�࣬�����Һ��c(Ca2+)=0.1mol/L������Һ��c(A2-)= ��
��ƽ�ⳣ�������˷����ϵ�Ŀ��淴Ӧ�ڸ������¶��½��еij̶ȣ�����ͬһ�����͵ķ�Ӧ��ƽ�ⳣ��Խ������Ӧ���еij̶�Խ��


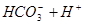









����������̼��ʹ�����ĵ���ƽ�ⳣ����д����������������������Ӧ�����ӷ���ʽ��
(1)������������ͨ�뵽������̼������Һ��___ _____________��
(2)������̼���ư���1�U1�����ʵ���֮��ǡ�÷�Ӧ_______________________________��