МвДҝДЪИЭ
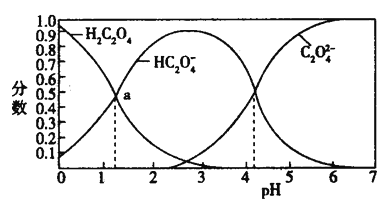
ЎҫМвДҝЎҝ¶МЦЬЖЪФӘЛШWЎўX ЎўY әНZ ФЪЦЬЖЪұнЦРөДПа¶ФО»ЦГИзұнЛщКҫЈ¬ХвЛДЦЦФӘЛШФӯЧУөДЧоНвІгөзЧУКэЦ®әНОӘ21 ЎЈ»ШҙрПВБРОКМвЈә
W | X | |||
Y | Z |
ЈЁ1Ј©X ФЪФӘЛШЦЬЖЪұнЦРөДО»ЦГОӘ_______ЎЈ
ЈЁ2Ј©ЛДЦЦФӘЛШјтөҘАлЧУөД°лҫ¶УЙҙуөҪРЎОӘ_____________ЈЁУГАлЧУ·ыәЕұнҙпЈ©ЎЈ
ЈЁ3Ј©WөДЧојтөҘЗв»ҜОпөДөзЧУКҪОӘ________________ЎЈ
ЈЁ4Ј©WЎўZ ЧоёЯјЫСх»ҜОпөДЛ®»ҜОпЛбРФҪПЗҝөДОӘ___________ЈЁМо»ҜС§КҪЈ©ЎЈ
ЈЁ5Ј©YөҘЦКУлFe2O3 ·ҙУҰДЬ·ЕіцҙуБҝөДИИЈ¬іЈУГУЪәёҪУёЦ№мЈ¬ёГ·ҙУҰ»ҜС§·ҪіМКҪОӘ____________________ЎЈ
ЈЁ6Ј©ПтКўУР3 mL јҰө°ЗеИЬТәөДКФ№ЬАпөОИлјёөОWөДЧоёЯјЫСх»ҜОпөДЛ®»ҜОпЕЁИЬТәЈ¬КөСйПЦПуОӘ________________________________________ЎЈ
ЈЁ7Ј©C3H7Z өДҪб№№јтКҪУР____________________ЎЈ
ЈЁ8Ј©ZX2ЖшМеКЗТ»ЦЦ№гЖЧЙұҫъПы¶ҫјБЎЈ№ӨТөЙПҝЙАыУГNaZX3әНNaZ ФЪЛбРФМхјюПВЦЖөГZX2 Н¬КұөГөҪZФӘЛШөДөҘЦКЈ¬ёГ·ҙУҰөДАлЧУ·ҪіМКҪОӘ_____________ЎЈ
Ўҫҙр°ёЎҝ өЪ2ЦЬЖЪ ўцAЧеЈЁ»төЪ¶юЦЬЖЪ ўцAЧеЈ© ClЁDЈҫN3ЁDЈҫO2ЁDЈҫAl3Ј« ![]() HClO4 2Al Ј« Fe2O3
HClO4 2Al Ј« Fe2O3 ![]() 2Fe Ј« Al2O3 ІъЙъ»ЖЙ«іБөн(»төӯ»ЖЙ«іБөн»тұд»Ж) CH3CH2CH2ClЎўCH3CHClCH3 2ClO3ЁD Ј« 2ClЁD Ј« 4HЈ« ЈҪ 2ClO2Ўь Ј« Cl2Ўь Ј« 2H2O
2Fe Ј« Al2O3 ІъЙъ»ЖЙ«іБөн(»төӯ»ЖЙ«іБөн»тұд»Ж) CH3CH2CH2ClЎўCH3CHClCH3 2ClO3ЁD Ј« 2ClЁD Ј« 4HЈ« ЈҪ 2ClO2Ўь Ј« Cl2Ўь Ј« 2H2O
ЎҫҪвОцЎҝУЙНјұнҝЙЦӘЈ¬ЛДЦЦФӘЛШФӯЧУөДЧоНвІгөзЧУКэЦ®әНОӘ21ЙиYөДЧоНвІгөзЧУКэОӘx Ј¬ФтУРx+x+2+x+3+x+4=21Ј¬ҪвөГx=3Ј¬WОӘNФӘЛШЎўXОӘOФӘЛШЎўYОӘAlФӘЛШЎўZОӘClФӘЛШЎЈЈЁ1Ј©X ОӘСхФӘЛШЈ¬ФЪФӘЛШЦЬЖЪұнЦРөДО»ЦГОӘөЪ2ЦЬЖЪ ўцAЧе»төЪ¶юЦЬЖЪ ўцAЧеЈ»ЈЁ2Ј©ЛДЦЦФӘЛШјтөҘАлЧУіэВИАлЧУ¶аТ»ёцөзЧУІгЈ¬°лҫ¶ЧоҙуЈ¬ЖдЛыАлЧУҫЯУРПаН¬өДөзЧУІгҪб№№Ј¬әЛөзәЙКэФҪҙуЈ¬°лҫ¶ФҪРЎЈ¬Фт°лҫ¶УЙҙуөҪРЎОӘClЁDЈҫN3ЁDЈҫO2ЁDЈҫAl3Ј«Ј»ЈЁ3Ј©WОӘөӘФӘЛШЈ¬ЖдЧојтөҘЗв»ҜОпNH3өДөзЧУКҪОӘ![]() Ј»ЈЁ4Ј©өӘЎўВИөДЧоёЯјЫСх»ҜОпөДЛ®»ҜОпHNO3ЎўHClO4ЛбРФҪПЗҝөДОӘHClO4Ј»ЈЁ5Ј©ВБөҘЦКУлFe2O3 ·ҙУҰДЬ·ЕіцҙуБҝөДИИЈ¬іЈУГУЪәёҪУёЦ№мЈ¬ёГ·ҙУҰ»ҜС§·ҪіМКҪОӘ2Al Ј« Fe2O3
Ј»ЈЁ4Ј©өӘЎўВИөДЧоёЯјЫСх»ҜОпөДЛ®»ҜОпHNO3ЎўHClO4ЛбРФҪПЗҝөДОӘHClO4Ј»ЈЁ5Ј©ВБөҘЦКУлFe2O3 ·ҙУҰДЬ·ЕіцҙуБҝөДИИЈ¬іЈУГУЪәёҪУёЦ№мЈ¬ёГ·ҙУҰ»ҜС§·ҪіМКҪОӘ2Al Ј« Fe2O3 ![]() 2Fe Ј« Al2O3Ј»ЈЁ6Ј©ПтКўУР3 mL јҰө°ЗеИЬТәөДКФ№ЬАпөОИлјёөОWөДЧоёЯјЫСх»ҜОпөДЛ®»ҜОпHNO3ЕЁИЬТәЈ¬·ўЙъСХЙ«·ҙУҰЈ¬КөСйПЦПуОӘІъЙъ»ЖЙ«іБөн»төӯ»ЖЙ«іБөн»тұд»ЖЈ»ЈЁ7Ј©C3H7Z ОӘC3H7ClЈ¬ЖдҪб№№јтКҪУРCH3CH2CH2ClЎўCH3CHClCH3Ј»ЈЁ8Ј©ClO2ЖшМеКЗТ»ЦЦ№гЖЧЙұҫъПы¶ҫјБЎЈ№ӨТөЙПҝЙАыУГNaClO3әНNaClФЪЛбРФМхјюПВЦЖөГClO2Н¬КұөГөҪВИЖшЈ¬ёГ·ҙУҰөДАлЧУ·ҪіМКҪОӘ2ClO3ЁD Ј« 2ClЁD Ј« 4HЈ« ЈҪ 2ClO2Ўь Ј« Cl2Ўь Ј« 2H2OЎЈ
2Fe Ј« Al2O3Ј»ЈЁ6Ј©ПтКўУР3 mL јҰө°ЗеИЬТәөДКФ№ЬАпөОИлјёөОWөДЧоёЯјЫСх»ҜОпөДЛ®»ҜОпHNO3ЕЁИЬТәЈ¬·ўЙъСХЙ«·ҙУҰЈ¬КөСйПЦПуОӘІъЙъ»ЖЙ«іБөн»төӯ»ЖЙ«іБөн»тұд»ЖЈ»ЈЁ7Ј©C3H7Z ОӘC3H7ClЈ¬ЖдҪб№№јтКҪУРCH3CH2CH2ClЎўCH3CHClCH3Ј»ЈЁ8Ј©ClO2ЖшМеКЗТ»ЦЦ№гЖЧЙұҫъПы¶ҫјБЎЈ№ӨТөЙПҝЙАыУГNaClO3әНNaClФЪЛбРФМхјюПВЦЖөГClO2Н¬КұөГөҪВИЖшЈ¬ёГ·ҙУҰөДАлЧУ·ҪіМКҪОӘ2ClO3ЁD Ј« 2ClЁD Ј« 4HЈ« ЈҪ 2ClO2Ўь Ј« Cl2Ўь Ј« 2H2OЎЈ

 ¶б№ЪСөБ·өҘФӘЖЪД©іеҙМ100·ЦПөБРҙр°ё
¶б№ЪСөБ·өҘФӘЖЪД©іеҙМ100·ЦПөБРҙр°ё РВЛјО¬РЎ№Ъҫь100·ЦЧчТөұҫПөБРҙр°ё
РВЛјО¬РЎ№Ъҫь100·ЦЧчТөұҫПөБРҙр°ёЎҫМвДҝЎҝОӘМҪҫҝNa УлCO2·ҙУҰІъОпЈ¬Ді»ҜС§РЛИӨРЎЧй°ҙПВНјЧ°ЦГҪшРРКөСйЎЈ
ТСЦӘЈәCO + 2Ag ( NH3) 2OH=2AgЎэ+( NH4 ) 2CO3 +2NH3

»ШҙрПВБРОКМвЈә
ЈЁ1Ј©РҙіцA ЦР·ҙУҰөДАлЧУ·ҪіМКҪ__________________________ЎЈ
ЈЁ2Ј©ТЗЖчX өДГыіЖКЗ_____________Ј¬B ЦРөДИЬТәОӘ________________ЎЈ
ЈЁ3Ј©ПИіЖБҝУІЦКІЈБ§№ЬөДЦКБҝОӘm1 g Ј¬Ҫ«СщЖ·Ч°ИлУІЦКІЈБ§№ЬЦРЈ¬іЖөГСщЖ·әНУІЦКІЈБ§№ЬөДЧЬЦКБҝКЗm2 g ЎЈФЩҪшРРПВБРКөСйІЩЧчЈ¬ЖдХэИ·ЛіРтКЗ________ЈЁМоұкәЕЈ©Ј»
aЈ®өгИјҫЖҫ«өЖЈ¬јУИИ bЈ®ПЁГрҫЖҫ«өЖ cЈ®№ШұХK1әНK2
dЈ®ҙтҝӘK1әНK2Ј¬НЁИлCO2ЦБE ЦРіцПЦ»лЧЗ
eЈ®іЖБҝУІЦКІЈБ§№Ь fЈ®АдИҙөҪКТОВ
ЦШёҙЙПКцІЩЧчІҪЦиЈ¬ЦұЦБУІЦКІЈБ§№ЬәгЦШЈ¬іЖөГЦКБҝОӘm3 g ЎЈ
ЈЁ4Ј©јУИИУІЦКІЈБ§№ЬТ»¶ОКұјдЈ¬№ЫІмөҪТФПВПЦПу
ўЩ ДЖҝйұнГжұдәЪЈ¬ИЫИЪіЙҪрКфРЎЗтЈ»
ўЪ јМРшјУИИЈ¬ДЖСёЛЩИјЙХЈ¬ІъЙъ»ЖЙ«»рСжЎЈ·ҙУҰНкИ«ә󣬹ЬЦРУРҙуБҝәЪЙ«ОпЦКЈ»ўЫ F ЦРКФ№ЬДЪұЪУРТш°ЧОпЦКІъЙъЎЈ
ІъЙъЙПКцўЪПЦПуөДФӯТтКЗ____________________________________ЎЈ
ЈЁ5Ј©МҪҫҝ№ММеІъОпЦРФӘЛШNa өДҙжФЪРОКҪ
јЩЙиТ»ЈәЦ»УРNa2CO3Ј»јЩЙи¶юЈәЦ»УРNa2O Ј»јЩЙиИэЈәNa2OәНNa2CO3ҫщУР
НкіЙПВБРКөСйЙијЖЈ¬СйЦӨЙПКцјЩЙиЈә
ІҪЦи | ІЩЧч | ҪбВЫ |
1 | Ҫ«УІЦКІЈБ§№ЬЦРөД№ММеІъОпИЬУЪЛ®әу№эВЛ | јЩЙиТ»іЙБў |
2 | НщІҪЦи1ЛщөГВЛТәЦР___________________________ | |
3 | _____________________________________________ |
ЈЁ6Ј©ёщҫЭЙПКцКөСйПЦПуј°ПВұнКөСйКэҫЭЈ¬РҙіцNa УлCO2·ҙУҰөДЧЬ»ҜС§·ҪіМКҪ___________ЎЈ
m1 | m2 | m3 |
66.7g | 69.0g | 72.1g |
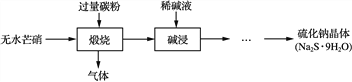
ЎҫМвДҝЎҝДі№Өі§¶Ф№ӨТөОЫДаЦРөДCrФӘЛШ»ШКХУлФЩАыУГөД№ӨТХИзПВЈЁТСЦӘБтЛбҪюИЎТәЦРөДҪрКфАлЧУЦчТӘКЗCr3+Ј¬ЖдҙОКЗЙЩБҝөДFe2+ЎўFe3+ЎўAl3+ЎўCa2+ЎўMg2+)Јә

Іҝ·ЦСфАлЧУіЈОВПВТФЗвСх»ҜОпРОКҪіБөнКұИЬТәөДpHјыПВұн:
СфАлЧУ | Fe3+ | Fe2+ | Mg2+ | Al3+ | Cr3+ |
ҝӘКјіБөнКұөДpH | 1.9 | 7.0 | ЎӘЎӘ | ЎӘЎӘ | ЎӘЎӘ |
іБөнНкИ«КұөДpH | 3.2 | 9.0 | 11.1 | 8 | 9ЈЁ>9ИЬҪвЈ© |
ЈЁ1Ј©КөСйКТУГ98%ЈЁГЬ¶ИОӘ1.84gЎӨcm-3)өДЕЁБтЛбЕдЦЖ200 mL 4.8 molЎӨL-1өДБтЛбИЬТәЈ¬ЕдЦЖКұРиТӘБҝИЎ98%өДЕЁБтЛбөДМе»эОӘ_______ mLЈЁұЈБфРЎКэөгәуТ»О»РЎКэЈ©Ј¬ЛщУГөДІЈБ§ТЗЖчіэЙХұӯЎўІЈБ§°фЎўБҝНІәНЛбКҪөО¶Ё№ЬНвЈ¬»№Ри_______ЎЈ
ЈЁ2Ј©№эВЛІЩЧчКұЈ¬РиТӘ¶ФіБөнҪшРРПҙөУЈ¬ПҙөУіБөнөД·Ҫ·ЁКЗ_______ЎЈ
ЈЁ3Ј©јУИлH2O2өДЧчУГТ»·ҪГжКЗСх»Ҝ+3јЫCrК№Ц®ЧӘұдіЙ+6јЫCrЈЁCrO42-»тCr2O72-Ј©Ј¬ТФұгУЪУлФУЦКАлЧУ·ЦАлЈәБнТ»·ҪГжКЗ_______ЎЈЈЁАлЧУ·ҪіМКҪұнКҫЈ©
ЈЁ4Ј©өчҪЪИЬТәөДpH=8іэИҘөДФУЦКАлЧУКЗ_______ЎЈ
ЈЁ5Ј©ДЖАлЧУҪ»»»КчЦ¬өДФӯАнОӘЈәMn++nNaRЎъMRn+nNa+Ј¬ұ»Ҫ»»»өДФУЦКАлЧУКЗ_______ЎЈ
ЈЁ6Ј©НЁSO2ЖшМеКұЈ¬»№Фӯ№эіМ·ўЙъТФПВ·ҙУҰЈЁМоРҙИұПоОпЦКІўЕдЖҪЈ©Јә
____ Na2Cr2O7+ SO2+ = Cr(OH)(H2O)5SO4+ Na2SO4ЎЈ