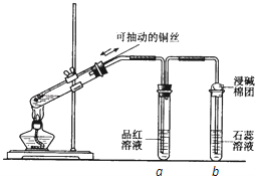
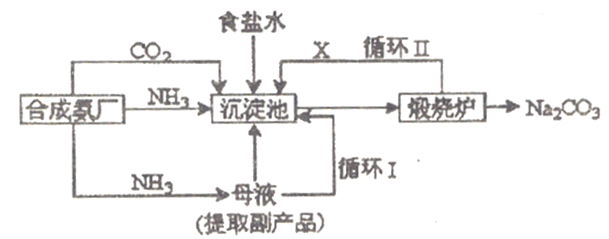
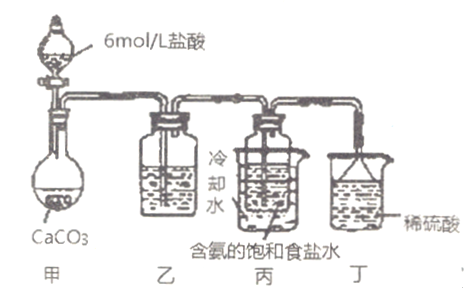
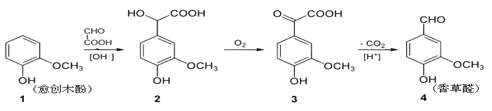
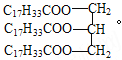��Ŀ����
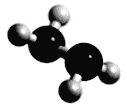
����Ŀ��������N2H4ΪҺ�壬����Ϊ�����������ȼ�ϣ���������N2O4������Ӧ��2N2H4 + N2O4 ![]() 3N2 + 4H2O���������ģ������ͼ��ʾ��NAΪ�����ӵ�������ֵ������������ȷ����
3N2 + 4H2O���������ģ������ͼ��ʾ��NAΪ�����ӵ�������ֵ������������ȷ����
A. 32 g N2H4�к��й��õ��Ӷ���Ϊ6 NA
B. ��״���£�22.4L N2H4�к��еķ�����ΪNA
C. N2H4��N2O4��ϵ������ʵ���Ϊ1molʱ������ԭ����Ϊ4 NA
D. ������Ӧ����1mol N2H4��ת�Ƶ�����Ϊ4 NA
���𰸡�D
��������A��32g N2H4�����ʵ���Ϊ1mol��ÿ����ԭ���γ��������ۼ��������й��õ��ӶԵ���ĿΪ5NA��ѡ��A����B����״���£�N2H4ΪҺ̬��22.4L N2H4�к��еķ���������NA��ѡ��B����C��ÿ�����Ӿ���2����ԭ�ӣ���N2H4��N2O4��ϵ������ʵ���Ϊ1molʱ������ԭ����Ϊ2 NA��ѡ��C����D����Ӧ2N2H4 + N2O4 ![]() 3N2 + 4H2O��N2H4�е�Ԫ�ش�-2�۱�Ϊ0�ۣ�����1mol N2H4��ת�Ƶ�����Ϊ4 NA��ѡ��D��ȷ����ѡD��
3N2 + 4H2O��N2H4�е�Ԫ�ش�-2�۱�Ϊ0�ۣ�����1mol N2H4��ת�Ƶ�����Ϊ4 NA��ѡ��D��ȷ����ѡD��

��ϰ��ϵ�д�
 ��ĩ�����ϵ�д�
��ĩ�����ϵ�д�
�����Ŀ